In Harry Potter and The Deathly Hallows Part 2, when Harry, Ron, and Hermione went to Lestrange Vault they did not know all of the treasure inside was cursed. Every time you touched one item, it multiplied into four additional items. Before long they are nearly drowning in an-ever multiplying flood of treasure. Until they finally decide to stop moving.
Now the entire world finds itself in a similar situation. The coronavirus, or COVID-19, is multiplying at an exponential rate. This global pandemic first consumed China and South Korea, then Italy and the rest of Europe, the Middle East, and now is spreading throughout the U.S. As a result, government organizations, doctors, and scientists are telling people to stop moving.
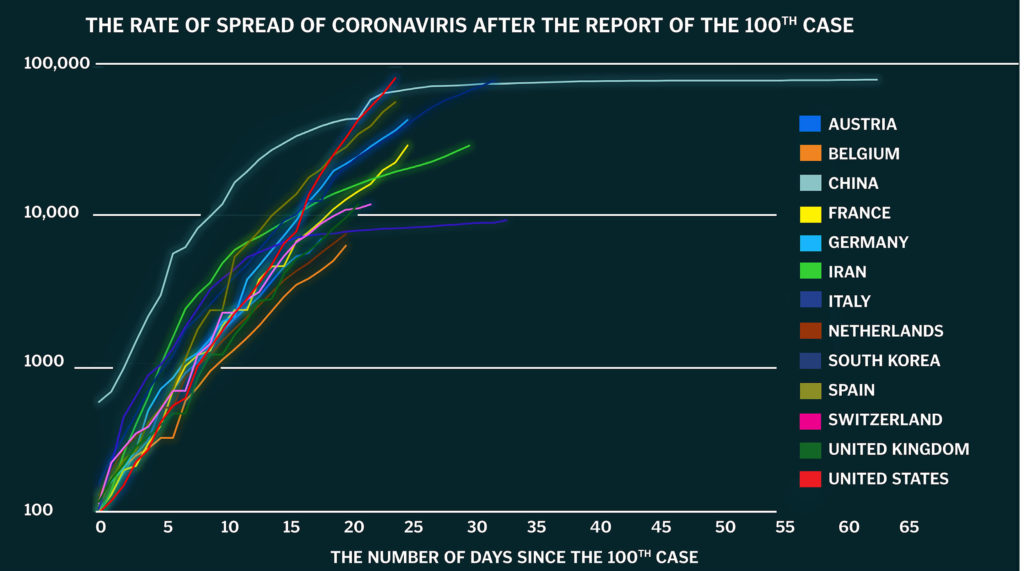
Borders are closed. Airports are shut down. All but eight states in the U.S. (Arkansas, Iowa, Nebraska, North Dakota, South Dakota, South Carolina, Utah, and Wyoming) have issued stay-at-home orders. Meanwhile, the CDC is still encouraging social distancing nationwide and recommending people remain six-feet apart and not to gather in groups larger than 10.
 And yet, the life sciences industry simply can’t stop moving. The world can’t afford it. Not only are manufacturers working tirelessly to develop treatments and vaccines for COVID-19, but these companies must also continue to engage with HCPs and patients to ensure all of their needs (COVID-19-related or not) are still being met. And while in-person engagements are now off the table, life sciences marketers and their partners are finding plenty of ways to ensure crucial information is still getting to the people who need it.
And yet, the life sciences industry simply can’t stop moving. The world can’t afford it. Not only are manufacturers working tirelessly to develop treatments and vaccines for COVID-19, but these companies must also continue to engage with HCPs and patients to ensure all of their needs (COVID-19-related or not) are still being met. And while in-person engagements are now off the table, life sciences marketers and their partners are finding plenty of ways to ensure crucial information is still getting to the people who need it.
Conferences Are Cancelled, But Not Completely Gone
Every conference and medical congress until May, if not June, has already been cancelled, postponed, or converted to a virtual meeting. But that doesn’t mean marketers still can’t reach the audiences that attend these meetings.
“Several congresses like the American College of Cardiology’s World Congress of Cardiology are converting to free virtual access programs allowing remote access to pre-recorded late-breaker presentations, moderated discussions, and virtual release of poster presentations which could in fact lead to greater exposure of new emerging data. Additionally, live and on-demand peer-to-peer education will be added to help supplement tele- and virtual-detailing from a medical marketing perspective, because the persistent cardiovascular risk of patients remains,” says Andrew J. Watson, Senior Director, Marketing, Amarin Pharma, Inc. “And while I believe there is no true substitute for pressing the flesh with opinion leaders, advocacy groups, and key customers, this black swan event will undoubtedly help to establish a new normal with respect to virtual medical meetings going forward.”
Agencies are also exploring ways to help clients move the conference experience online. For example, Paul Hagopian, a founder at SPLICE Agency, says they are building virtual reality or augmented reality booths, presentations, and posters—essentially an entire immersive infrastructure that can even be customized to the users. These virtual worlds can be navigated similarly to a live conference, done more like a choose your own adventure experience, sent out as email blast, or a print piece with an AR component. However, he warns this approach won’t work for everyone.
“Those virtual experiences take time to develop properly and agencies will have to be nimble, because if it takes you six months to develop, then we might be back to having live events by then,” Hagopian offers. “Being that cost and speed of response is paramount, some of our clients are leveraging their existing materials and going back to basics blasting their audiences with direct mail and print campaigns. You’re not necessarily getting the immersive experience of AR/VR, but that’s probably your fastest way to respond to the current situation.”
Reimagining Rep Visits and Other Face-to-Face Meetings
Life sciences companies are taking their sales reps off the road, either due to their own safety concerns or the fact hospitals won’t let them in at this time. In fact, 87% of surveyed companies say they have completely pulled reps from the field, according to a TGaS Advisory Insights report from March 17. To still make sure they are reaching their HCPs, 61% said they have increased their use of virtual meetings, 32% increased tele-sales, and 19% have modified their sampling program to ensure adequate supply for HCPs.
 “If we can’t reach doctors live then we are utilizing teledetails, using things like Veeva-approved emails to send out invitations for doctors to join a webinar, conference, or something similar,” says Kendra Fanara, Executive Director, Marketing Operations, Sunovion. “And because COVID-19 is already a disruption, you don’t want to accrue any more disruption. So the faster companies can turn to digital engagements, the better off we are all going to be.”
“If we can’t reach doctors live then we are utilizing teledetails, using things like Veeva-approved emails to send out invitations for doctors to join a webinar, conference, or something similar,” says Kendra Fanara, Executive Director, Marketing Operations, Sunovion. “And because COVID-19 is already a disruption, you don’t want to accrue any more disruption. So the faster companies can turn to digital engagements, the better off we are all going to be.”
As a provider of technology used for teledetails, Veeva is making its Engage Meeting platform available to all of its current Veeva CRM customers for no additional license fee through the end of August. While you have to be a Veeva CRM customer to use this feature, any new customers will also be able to take advantage of this offer. So far, Veeva has already provisioned about 150,000 Veeva CRM Engage licenses since the start of the free offering. Engage Meeting allows reps or medical science liaisons to interact with HCPs remotely, both through audio and video, while also sharing any scientific materials or information in a regulatory compliant fashion.
“One company in China turned on this capability and eight days later they were able to start remotely interacting with customers,” says Paul Shawah, Sr. VP of Commercial Strategy, Veeva. “There’s not a whole lot of training because they’ve been training for this already—it’s still face-to-face, just a different medium.”
It’s not just rep visits moving virtual. Chris Sweeney, Chairman and Chief Executive Officer of HealthSTAR Communications, a company that specializes in live and digital peer-to-peer engagement, says his company is currently in the midst of converting 75% of its scheduled live events for clients to virtual engagements.
“HealthSTAR has reinforced our proven virtual engagement infrastructure and virtual solutions, providing clients and the industry with the opportunity to convert live meetings to virtual engagements for tactics such as speaker programs/trainings and advisory boards,” Sweeney explains. “And HealthSTAR’s virtual capabilities are not just for pharma companies, we have spoken to other agencies about utilizing our technology.”
But as companies turn to more non-personal promotion (NPP) in the short term, the question is whether this could impact how companies operate even after the COVID-19 crisis has passed.
“I think some significant changes will come out of this, including a stronger reliance on NPP,” says Gary McWalters, President of TGaS Advisors, a division of Trinity. “Pharma has been doing NPP for a number of years, but the industry has never been willing to do the type of A/B testing necessary to prove whether these tactics are truly effective.”
And if NPP does prove to be as effective, what could that mean for reps?
“We had already been living in a world where access for reps has been tapering, and this current situation is taking what was a tapered trend and all of a sudden turning it up to 11,” Hagopian says. “So after this situation is over, it will be really interesting to look at the balance between live rep engagement and NPP and reevaluate it. With that said, the rep engagement with the customer will always have value, but in my opinion, the integration of marketing technology during those engagements will drive a more enduring brand footprint and stronger ROI. The next frontier for customer engagement will involve reps truly embracing and leveraging technology, maximizing their value as the ‘customer service face’ to their customers.”
Connecting at the Point of Care
As doctors’ offices and hospitals start to become flooded with patients who have COVID-19, believe they have it, or are just dealing with other illnesses, the point of care becomes an important place to distribute the latest information.
For example, OptimizeRx is pushing information from the CDC about COVID-19 to thousands of HCPs nationwide directly within the EHR. The company segmented patient populations by age, gender, comorbidities, diseases, etc., to identify at-risk populations per CDC guidelines in order to ensure the information is getting to HCPs who are engaging with patients that meet that criteria. Additionally, with reps’ access limited, OptimizeRx continues to distribute relevant information and educational materials from life sciences companies through its EHR platform across specialties.
Meanwhile, in waiting rooms, other point of care vendors such as Mesmerize, Outcome Health, and PatientPoint are developing CDC- and WHO-sourced information about COVID-19 and displaying it on wallboards, posters, televisions, and tablets. And according to Mesmerize CEO Gregory Leibert, while this information is being prioritized, other materials from life sciences marketers are continuing to be displayed on their network.
“Our content management system software allows us to weigh media and balance plays, and we’ve assigned the COVID-19 media a higher value than our standard health and wellness content,” Leibert says. “This allows us to have maximum exposure for the COVID-19 content, while still meeting the contractual obligations to our advertising partners and providing space in our loop for other important location-specific patient education.”
Even in exam rooms, companies are making adjustments to ensure patients can view information safely. PatientPoint has temporarily converted its Interactive Exam Room Devices into a “touchless” platform by allowing patients to scan a QR code so that they can view the content on their own phones.
Finding Ways to Reach Patients
Life sciences companies that like to engage with patients in-person through speaker programs, advisory boards, local patient outreach programs (LPOPs), and other events must also change their tactics.
Blake Shewey, EVP of Snow Companies, an agency that specializes in patient engagement, says their company always operates with caution in order to safeguard its patients and is experienced in working with patient communities who cannot participate in live events, even before this, such as those with cystic fibrosis. For example, advisory boards can be done in a virtual environment on a compliant platform and their agency’s Patient Ambassadors can share their stories through video, audio, or a virtual meeting. Additionally, when it’s safe for smaller groups to meet, it can be broadcast so others can join remotely.
Jenn Etheridge, Senior Marketing Manager at Genentech, says that while they are certainly looking at various ways to connect with patients virtually, it will depend on the therapeutic area.
“We’re still evaluating whether it would work in idiopathic pulmonary fibrosis (IPF) because that’s a scenario where people tend to not be as technologically savvy and you don’t want people to miss out on education because of that,” she explains. “So we’ve asked our Clinical Coordinators who educate patients to try and do things virtually or by phone because we recognize that patients still have questions and need information about their medication. So for the duration of this, we’re doing this by web conference or phone while adhering to proper compliance by providing important safety information and also mailing the USPI as a follow-up.”
Mobile is another way companies are getting information to patients. In addition to their EHR platform, OptimizeRx is also leveraging its patient engagement expertise during the COVID-19 crisis. The company continues to deliver patient engagement programs for life sciences companies on its mobile platform and has additionally launched a free alert so that consumers can receive CDC info and alerts about COVID-19 by texting VIRUS to 55150.
And Omri Shor, CEO and Co-founder of Medisafe, a digital therapeutics company for medication management solutions, says they are now actively working with their clients/partners to incorporate digital medication management support solutions, at no additional cost, to lighten the burden on their patient support resources. The company is also scaling a package to meet industry need for digital companion solutions to bridge the “patient distancing needs” as well as offering online pharmacy refill services.
A Time for Innovation
Marketers and their partners are also working on other innovative solutions to help limit the spread of this pandemic. For instance, Klick Health has already supported the Dock Health implementation of COVID-19 checklists in collaboration with Dr. Michael Docktor and the Boston Children’s Team and offered up some team members to support the data visualization efforts being led by Dr. John Brownstein’s Harvard team working in collaboration with Mapbox to make www.healthmap.org/covid-19.
The company is also exploring other projects such as whether Machine Learning can distinguish between COVID-19 coughs vs. other coughs in a reliable manner, and then be used in combination with a thermometer and a symptom checker as a triage tool. One of Klick’s data scientists even built a prototype for a “don’t touch my face” app that can utilize a person’s webcam, at a time when more people are working virtually, to help people break an all-too-common habit that they are now being advised against.
And most recently, the company made a public call to action for ideas on how to use digital health to manage and contain COVID-19. Anyone is free to submit their idea at https://COVID19.klick.com. Klick will select the best ideas and help bring them to life—at no cost—and the solution will be made available to the public for free.
“Our vision is to select a couple of projects we believe will make a difference and then focus,” says Klick Health President Lori Grant. “We’re evaluating the ideas using a variety of important criteria, such as: Can the idea have impact; is it scientifically plausible; and can it be implemented relatively quickly without regulatory hurdles? This is about everyone working together and recognizing that this is what’s required to protect our parents, loved ones, the vulnerable, as well as the heroic healthcare professionals and first responders.”