DC Dispatch: Call for Obesity Prevention Tax Codes
Led by the Campaign to End Obesity Action Fund, 22 concerned groups sent a letter to the leaders of congressional tax-writing committees urging them to embrace tax policy solutions to help control the U.S. obesity epidemic.
The NAACP, National Association of County and City Health Officials, National Hispanic Medical Association and others pressed leaders of the Senate Finance Committee and House Ways & Means Committee to advance tax incentives to improve access to resources including grocery stores, safe recreational spaces and other infrastructure needed to bolster healthy living. With ongoing discussions in Congress about the tax reform needed, the groups urged policymakers to craft tax measures that will:
- Urge private interests to increase access to healthy, affordable foods in economically disadvantaged communities.
- Provide increased access to safe recreational spaces.
- Support economically disadvantaged individuals in efforts to adopt lifestyle choices that can reverse or prevent obesity.
- Prompt businesses to invest in tools and resources to encourage healthy lifestyle choices.
- Be targeted to benefit those individuals and communities most at risk for obesity.
In total, the obesity epidemic (affecting nearly 100 million adults and children) costs American taxpayers nearly $200 billion each year in healthcare expenditures alone, so targeted tax policies that curb healthcare spending through reductions in obesity rates can generate meaningful long-term savings for the federal budget.
Trend Setting: Physicians Want Out of Stagnant Pay Trend
With the current state of fee-for-service reimbursement in decline, physicians nationwide are exploring new ways to keep their medical practices thriving. Reimbursement levels for new and existing patient visits declined or remained flat in 2013, according to the annual Fee Schedule Survey by Physicians Practice, the leading online community for practice management professionals.
Since 2001, pay for physicians has stagnated nationwide. Data from 2013 indicates relatively flat reimbursements for existing patient visits, but decline in all the categories of new patient visits, including a 21% decrease in the highest complexity code (99205) used by medical practices. The data reflects 2,619 medical practices across the country.
To boost practice revenue, physicians are looking at new strategies, including partnering with other physicians and health systems, developing accountable care organizations (ACOs), participating in pay-for-performance programs or creating Patient-centered Medical Homes, all of which are getting increased attention due to their inclusion in the Affordable Care Act.
Patient Pages: Digital Portal for Childhood Cancer Survivors
Akron Children’s Hospital, with funding from Hyundai Hope on Wheels, is giving childhood cancer survivors the resources they need to live a healthy adult life. With a free, personalized mobile app and website, CancerLateFX (www.CancerLateEffects.org), childhood cancer survivors can get educational information, tips, tools and resources to keep them informed and healthy throughout their adult lives.
Instead of providing general health guidelines, the app generates content based on a person’s symptoms and medical history. CancerLateFX also uses the survivor’s cancer type and specific medications/treatments to introduce goals, prevention tips and talking points that can be further explored with the user’s current healthcare provider.
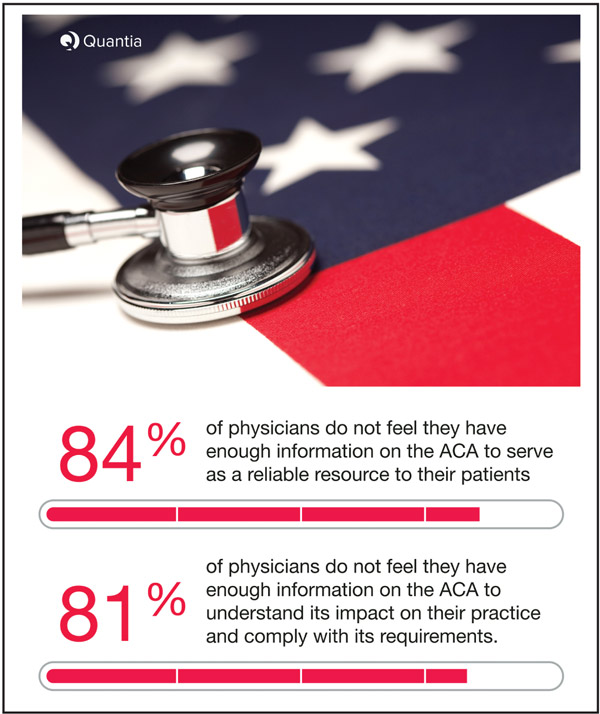
Doctor Docs: Physicians Left Out of ACA Conversation?
One of the largest online learning networks for physicians, QuantiaMD, recently completed a member poll regarding the implementation of the Affordable Care Act (ACA). It gathered responses from 1,265 physicians across the country, and the results are eye opening. Despite millions of enrollees, patients and physicians, according to the poll, remain confused about the law, a problem because many individuals turn to their physicians as a primary source of information on healthcare policy. The poll revealed:
- 84% of physicians said they did not feel they had enough ACA information to serve as a reliable resource for their patients.
- 81% of physicians also felt that they don’t have enough information to understand the ACA’s impact on their practice and how to comply with its requirements.
- 35% of physicians say that there aren’t any reliable sources of information, when asked where they get the most reliable information about the ACA.
In response to the findings of its poll, Quantia, Inc.’s Editor-in-Chief, Mike Paskavitz says, “This poll proves how physicians have been left out of the healthcare reform process, and validates the importance of Affordable Care Act curriculum.” Quantia, Inc., the creator of QuantiaMD, is developing such a curriculum for the one in three physicians nationwide who visit their site each quarter.
FDA Update
FDA Regulates Study Aimed at Treating Sensorineural Hearing Loss
The world’s largest newborn stem cell company, Cord Blood Registry, announced the start of a FDA-regulated study conducted at Florida Hospital for children in Orlando to investigate using a child’s stem cells from their stored umbilical cord blood as a treatment for acquired sensorineural hearing loss, the most common type of hearing loss in children.
Drug-device Combination Seeks Approval
The FDA assigned a user fee goal date of April 15, 2014 for MannKind Corporation’s Afrezza (insulin human [rDNA origin]) Inhalation Powder. MannKind is seeking FDA approval of Afrezza, an inhaled and ultra rapid-acting mealtime insulin therapy, to improve glycemic control in adult patients with type 1 or type 2 diabetes. The drug-device combination product delivers insulin using a small, discreet and easy-to-use inhaler.
New Approvals
Alere Connect is granted a 510(k) market clearance for OTC use of the Alere Homelink, part of a series of connected device technologies designed to allow patients self-testing at home abilities that deliver data quickly to clinical systems accessed by their healthcare providers.
Varian Medical systems received FDA 510(k) clearance for its ProBeam Proton Therapy System, which gives clinicians options for precise dose delivery to minimize the dose to healthy tissue while delivering proton therapy treatments for cancer.
The FDA approved Actavis’ generic version of Micardis, an angiotensin ll receptor blocker indicated for the treatment of hypertension, to lower blood pressure and reduce cardiovascular risk in patients who can’t take ACE inhibitors.
Vanda Pharmaceuticals’ new orphan drug Hetlioz, (tasimelteon), is the first FDA approved medication in the U.S. to treat non-24-hour sleep-wake disorder, a condition that causes problems in the sleep cycles of blind people. The disorder (FDA classified as a rare disease), affects about 800,000 Americans and causes difficulty falling or staying asleep, which can lead to a reversal in sleep patterns.