In order for marketers to be effective in driving business growth they need meaningful new products in the pipeline to bring to market. A new innovation from a company outside pharma may actually help to speed up the drug discovery process and shore up the pipeline.
The path to find new drugs is arduous and messy and companies continue to look for novel ways to find new medicines, ranging from partnerships with venture capital and drug discovery firms to restructuring internal R&D operations. Depending on the molecule, it can cost $15 million to $50 million to take a promising idea through proof-of-concept testing in humans and along the way the process is inefficient and rife with setbacks.
The tools and processes used in drug discovery have a profound effect on the outcome. Recently, an innovative new assay tool in fluorescent detection of protease enzyme activity from Enzium caught my eye. Enzium is a biotools company spun out of a technology licensed from Carnegie Mellon University and developed by Crystal Falco and Dr. Peter Berget (www.enziumlabs.com).
“We initially started developing the technology by special request from NIH to develop tools to make imaging cell modification processes (cleavage, phosphorylation, pH changes, etc.) easy,” explains Falco. “After speaking to end-users to gain input on how to best engineer these things we realized we were able to use our technology in many different applications beyond basic fluorescence imaging and we could provide products that could allow researchers to answer questions that they couldn’t answer before.”
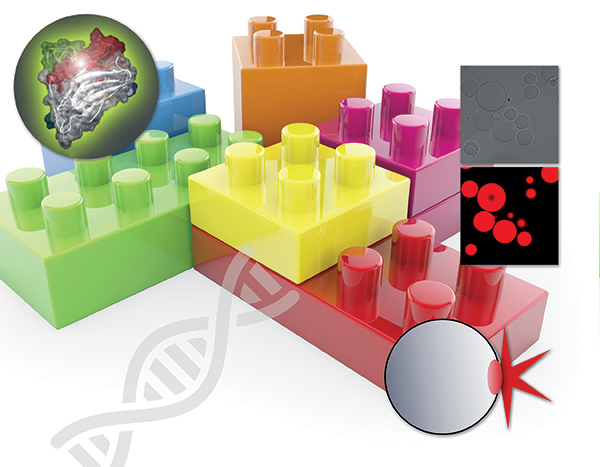
Enzium specializes in off-the-shelf and custom reagents kits that are more selective and easier to use for protease activity detection than organic chemical-based technologies. Much like a set of molecular Legos, their reagent platform allows a product development team to quickly pop amino acid protease recognition sequences—of any length—in and out of their reagents. This protein-based system not only allows the user to engineer in longer amino acid recognition sites than competing technologies, but it allows folding into tertiary and quaternary structures often preferred by proteases, naturally building in more selectivity for detecting specific proteases.
There are over 500 different proteases in the human body and thousands in the microbial world. Proteases cleave to other proteins and are a major class of enzyme target for drug discovery due to the roles they play in many disease pathways, such as cancers, neurological diseases, cardiopulmonary diseases, immune response and inflammatory disorders, along with many more. Often times several proteases of the same enzyme family play roles in a particular disease but current technology does not provide an effective way to selectively decipher when these proteases become active/inactive. Due to the way Enzium has engineered its reagents, enhanced selectivity for protease activity is built in.
Let’s take matrix metalloproteinases (MMPs) as an example. When a third-party lab tested a well-known commercial kit advertised to be selective for the detection of MMP-2 activity, they found that this reagent was also activated by MMP-14. Both MMPs play a role in cancer cell migration and inflammation. If developing a drug against MMP-2, using this reagent kit, the end-user would have no idea if their drug were actually regulating MMP-2 or MMP-14. The third-party lab tested Enzium’s MMP-2 reagent and found that it was only activated by active MMP-2. According to Dr. Peter Berget, “There was no cross reactivity with any of the other MMPs in that enzyme family. They also found this increased selectivity to be true for Enzium MMP-9, MMP-14 and MMP-25 reagents that were tested.”
Proteases require specific peptide sequences in order to recognize their targets. Some only require a few amino acids while others require hundreds of amino acids and/or tertiary and quaternary protein structure to identify targets for cleavage. Current technologies on the market are also limited by the length of the protease recognition sequence (about 20 amino acids or less) that can be built into a reagent. Since many proteases require longer peptide sequences for recognition, this limits research to a handful of well-characterized proteases. What is unique about the Enzium protein reagent technology is it allows for use of very long recognition sequences and opening up the potential to detect a larger breadth of proteases. Botulism neurotoxin protease is a good example. Though there are other reagents available to detect activity of this protease, it can only accommodate a 17 amino acid recognition sequence when the natural protease prefers a 230 amino acid sequence. Enzium has built a reagent with both the 17 amino acid sequence and the 230 amino acid sequence. They have demonstrated that the reagent with longer sequence is more sensitive allowing detection of picomolar levels of toxin enzyme.
Enzium is an example of a meaningful innovation coming from a company outside of the pharmaceutical industry. Their activity detection reagents can improve current drug screening efforts as well as provide the necessary data to progress a drug that has become stuck in the FDA’s approval process. The adoption of better tools to provide new information in the drug discovery process is one of the exciting ways to advance our progress to bring new medicines to the market.