The market of products that treat cardiovascular conditions is massive, with hypertension and cholesterol medications alone accounting for $34.5 billion in sales and almost 19% of all pharmaceutical prescriptions during the 12 months ending November 2011.
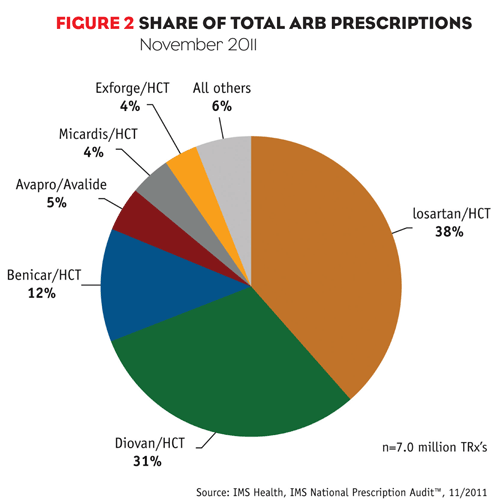
Hypertension is the top condition seen and treated by office-based physicians each year. Several different types of medications are used either alone or together to treat hypertension. The most popular class of drugs is angiotensin-converting enzyme (ACE) inhibitors, which accounted for 30% of the hypertension market’s 541 million prescriptions in the 12 months ending November 2011. Following ACE inhibitors were beta-blockers and calcium channel blockers (CCB) with 24% and 18% of market prescriptions, respectively.
ARBS
The fourth-most popular type of hypertension therapy, angiotensin receptor blockers (ARBs) and drugs that combine an ARB with a generically available CCB or other type of antihypertensive, accounted for 15% of market prescriptions—but a staggering 53% of sales in the year ending November 2011. Why are the market shares so off balance? Most ARBs are brands; the other classes are dominated by much less expensive generics.
Until April 2010, ARBs were without direct competition from generics. That month, losartan and losartan HCTZ, generic versions of Cozaar and Hyzaar, became available. For years before that, Novartis’ Diovan and Diovan HCT ranked first in ARB prescriptions month after month because of strategic and successful managed-care contracting and significant investments in promotion.
Novartis’ strategy included contracting with managed-care plans to place Diovan on tier two of their benefit designs (the preferred tier for brands) with only one or two other ARBs. After securing these types of contracts, Novartis then promoted Diovan’s preferred status and lower patient co-pays to physicians. In a class with little perceived product differentiation, lower co-pays and little payer issues at the pharmacy made Diovan an attractive option for physicians.
These contracts were positive for managed-care plans; Diovan held significant market share and accounted for high prescription volumes, motivating payers to continue to contract similarly with Novartis. Diovan benefited because the contracted limit to the number of ARBs that could share tier-two status stifled the growth of other brands. Benicar/Benicar HCT has made some inroads with managed-care plans and physicians, but has not yet approached Diovan’s prescription volumes.
In May 2011, more than a year after losartan/losartan HCTZ became available, prescriptions for the generics exceeded Diovan/Diovan HCT prescriptions for the first time. Most recently, in November 2011, losartan/losartan HCTZ accounted for 38% of ARB prescriptions. Diovan/Diovan HCT accounted for 31% and Benicar/Benicar HCT for 12%.
Initially, the shift to losartan happened at the pharmacy, as patients taking Cozaar and Hyzaar were easily switched from branded to generic therapies. More recently, the change is happening at physicians’ offices. New patients are being prescribed losartan/losartan HCTZ more than any other ARB. In November 2011, 57% of prescriptions for new-to-market patients were for losartan/losartan HCTZ. Diovan/Diovan HCT followed with just under 20%.
PATIENT COSTS
With generic competition, managed-care coverage and reasonable patient costs are more important for brands than ever before. As a generic, losartan/losartan HCTZ has the best managed-care coverage and lowest patient co-pays. In the year ending November 2011, the average patient out-of-pocket cost for a losartan/losartan HCTZ prescription was $12.09.
Diovan/Diovan HCT still enjoys solid managed-care coverage, especially in Medicare Part D plans. In the 12 months ending November 2011, the average patient out-of-pocket cost for a Diovan/Diovan HCT prescription was $35.22. In Medicare Part D, it was $29.99. The out-of-pocket cost for Diovan was the lowest of the branded ARBs. Benicar/Benicar HCT’s average out-of-pocket cost was $40.
Interestingly, although Diovan is more expensive than losartan, the percentage of submitted prescription claims that are not paid—either because the patient or payer rejects them—is comparable. In the year ending November 2011, 92% of submitted prescription claims for both Diovan and losartan were paid. Patients rejected 3.2% of Diovan prescriptions; payers rejected 4.7%. For losartan, patients rejected 3.4% and payers rejected 4.5% of submitted prescriptions. With cost the most common reason why patients reject a prescription, it is extremely positive for Diovan that its rejection rate isn’t higher than that for the generic. Benicar didn’t perform as well, with 3.9% of prescriptions rejected by patients and 6.4% rejected by payers.
Rejected claims cause problems beyond just the original lost business. In many cases, if a prescription is rejected, the pharmacy will try to replace the prescription by reaching out to the patient and physician. If physicians repeatedly are contacted over the same drug being rejected, they likely will decrease their prescribing of that medication.
COMBINATION PRODUCTS
Manufacturers know that in the cardiovascular market, cost and managed-care coverage can drive success. And, with concomitant drug usage across different classes, combination products are a way for manufacturers to get more mileage from their brands before generic equivalents surface. Most ARB combination products join a branded ARB with amlodipine, the generic equivalent of the calcium blocker Norvasc. The most successful of these combination products are Exforge and Exforge HCT, which join amlodipine and Diovan or amlodipine withDiovan and hydrochlorothiazide.
In November 2011, Exforge/Exforge HCT accounted for 4% of ARB market prescriptions. In addition to the patient benefit of fewer pills to take each day, Exforge is cheaper than individual prescriptions for the ingredient drugs. In the year ending November 2011, the average out-of-pocket cost for an Exforge prescription was $37.61. The combined cost of generic amlodipine ($7.66) and Diovan ($35.22) was over $5 more at $42.88.
In order for the remaining brands and combination products in the ARB market to continue to compete, they must maintain good managed-care coverage and marketing budgets sufficient to remind physicians of reasonable patient co-pays. Given the perception that drugs in this market are very similar, brands are at great risk of being replaced by the generically available ARBs.
FIGURE 1:
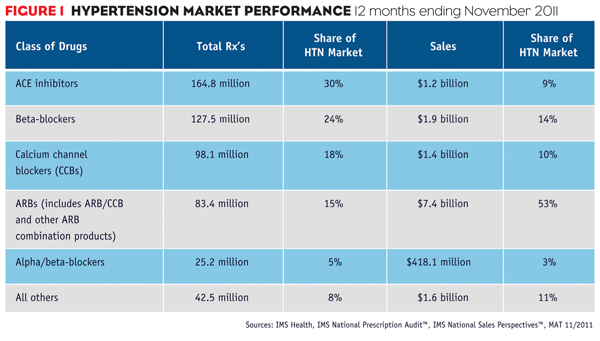
FIGURE 2: