Big Tech companies have mastered the consumer experience, enabling the likes of Apple, Google, and Amazon to evolve within a few short years from tiny Silicon Valley startups to trillion-dollar market cap behemoths. Through a robust combination of digital profiling, precision targeting, and dynamic engagement, they provide an unprecedented level of personalization, two-way communication, and real-time responsiveness that has transformed all of our lives.
Understanding the end-user is a prerequisite for success in most industries, arguably none more important than healthcare. But unlike consumers of media, commodities, or luxury goods—whose buyers consider shopping a flattering reflection of themselves and their values—patients are instead burdened by their disease, viewing treatment options as the only way to resume their everyday lives. The stakes couldn’t be any higher.
That makes the relatively slow incorporation of the patient voice into the clinical development and commercialization of drugs puzzling. Part of the challenge has been the paternalistic nature of the healthcare system itself, where physicians have historically been anointed sole decision-makers. But thanks to the rise of patient advocacy—largely driven by digital data sharing and social media—patients have an increasingly powerful voice in their own care.
Healthcare companies are now behooved to listen to that voice, and have already made great strides throughout the downstream marketing of post-launch treatments. Many of the same tools used by technology and media firms are now being applied by pharma and life sciences to better understand and empower patients to become stewards of their own healthcare journey, resulting in a progressively improving patient-brand relationship. While strides have been made on the commercial side, opportunities abound to bring the patient voice to the center of clinical drug development.
Getting it Right from the Start
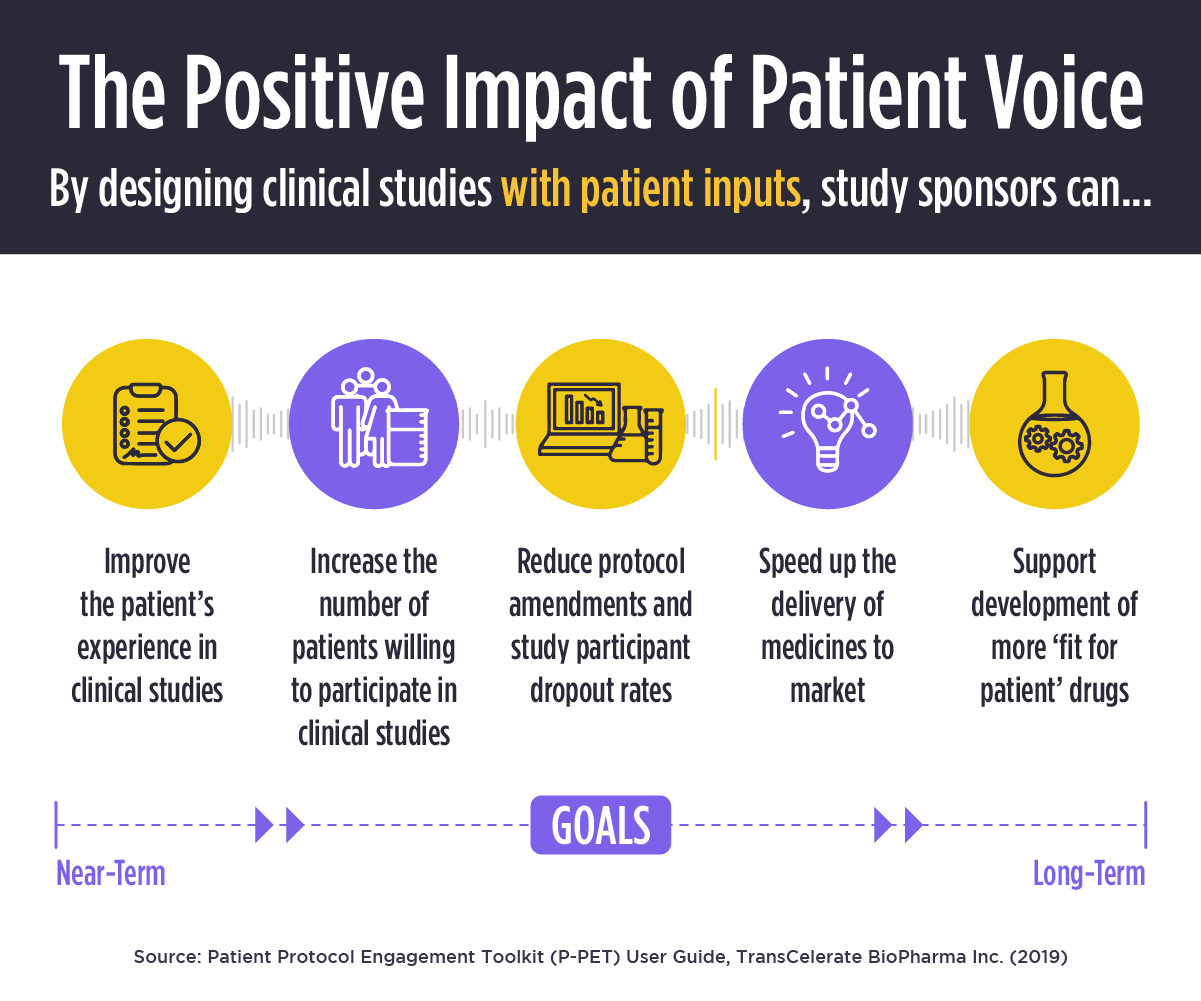
Key decisions made in the early stages of drug development cascade throughout the lifecycle of pharma brands, so gaining insights into patient needs is vital. The implications are consequential, both in terms of potentially costing pharma companies millions, and compromising the patient experience itself.
For example, lacking sufficient understanding of patient attitudes toward disease burden, a pharma company may design a clinical study that sufficiently meets its end-points, yet leads to approval of a drug with low adoption. Conversely, companies may fail to move forward with otherwise promising drug candidates due to questionable tolerability—yet if asked, patients would be willing to handle side effects in exchange for an improved overall quality of life.
Since the pharmaceutical industry has repeatedly developed products that fail to meet these critical patient needs, Congress and the FDA recently issued the Cares Act and extensive new guidance to ensure patient voices are heard at key touch points throughout clinical development. With a renewed focus on collecting comprehensive and representative patient input, fresh methods to identify relevance, and incorporating clinical outcomes assessments into endpoints for regulatory decision-making, patient insights are becoming increasingly influential.
Taken together, these governmental and industry-based initiatives have spawned a new era in patient-focused drug development (PFDD), uniquely positioning patients to inform the therapeutic context and make actionable recommendations. Although just beginning, PFDD has already systematized approaches to data collection, identified best practices, captured data on tradeoffs between benefits and risks, and analyzed input on communications preferences.
Hurdles nonetheless abound, many of them endemic to the pharmaceutical and healthcare industry. These include a patchwork of regulations and policies across a chaotic and evolving landscape; varied market needs from region to region, and country to country; failing to build an internal company culture conducive to patient-focused research; limited cross-functional collaboration; and hesitation on the part of executive leadership, often stuck in old habits.
Fortunately, the clinical process naturally lends itself to direct patient involvement—pharma simply needs to listen and more proactively participate. After all, patients are already recruited into clinical trials, share their feelings about the disease burden and their treatment journey with experts, and actively assist with the generation of evidence and actionable data. Qualitative interviews and the managing of patient thought leaders further enhance their burgeoning role.
But Pharma Needs to Listen
Effective inclusion begins with study design, where technologically facilitated qualitative research can extract the candid thoughts and feelings of patients with the goal of increasing their participation and retention. By better understanding end-users through patient-reported outcomes measures, researchers are able to create more patient-centric trials that produce increasingly meaningful results, ultimately approving safer and more effective drugs.
Past the clinical trial phase, improved market research methods can also further optimize every forthcoming step of drug development. From nomenclature to naming, branding to messaging, reimbursement to support and relationship management programs, tapping into the patient voice informs and empowers researchers, educators, marketers, and the vested healthcare professionals who discover, evaluate, and prescribe the approved drugs.
A compelling example of infusing the patient voice at the earliest stages of the development process was for Trikafta, the triple-combination treatment of cystic fibrosis from Vertex. A collaboration between the CF Foundation (that helped raise millions for research) and scientists, patients, and their families brought the patient voice into many aspects of the clinical process. This resulted in a breakthrough drug that launched months ahead of schedule.
 But challenges remain for clinical teams struggling for patient input, often employing legacy quantitative and qualitative market research methods that remain resource-intensive, inefficient, time consuming, expensive, and inflexible. Traditional approaches, including speaking with advocacy groups, convening patient ad boards, conducting phone interviews, and relying on the collective experience and intuition of brand teams can only go so far. Innovation is needed.
But challenges remain for clinical teams struggling for patient input, often employing legacy quantitative and qualitative market research methods that remain resource-intensive, inefficient, time consuming, expensive, and inflexible. Traditional approaches, including speaking with advocacy groups, convening patient ad boards, conducting phone interviews, and relying on the collective experience and intuition of brand teams can only go so far. Innovation is needed.
Embracing Patient-Focused Drug Development
Exploring novel creative and agile methods are therefore necessary to ensure patient insights are captured and resonate. The good news is that many of the same tools used by Big Tech for commercialization—alongside proven patient outreach techniques—can be applied to market research conducted at earlier clinical development phases. Let’s consider each step of the data collection, processing, and reporting cycle, and see how next-generation researchers are gaining transparency into the hearts and minds of patients across numerous disease states.
Data collection tools are now able to capture every nuance of a patient’s cognitive response and emotional intent through voice and other modalities. By recording frank patient reactions to creative and messaging stimuli, along with detailed, often stream of consciousness answers to open-ended questions, researchers can then process gigabytes of data with more depth and sophistication than through limiting legacy surveys and questionnaires.
The data processing demands of such layered input is met through a combination of AI/machine learning technology and human-powered analysis. Cutting edge semantic analysis software known as GPT-3 is also being used to rapidly search through and extract relevant content, laden with meaning and emotional context. The result is a dynamic database capable of answering secondary and tertiary research queries, diving deep into the patient’s world.
Without the reporting of actionable market research intelligence, the entire effort would be for naught. Here the dynamic, data-driven dashboards common to digital platforms are helping clinical researchers better understand and contextualize patient feedback and sentiment. Actionable patient-driven recommendations for the optimization of trial designs, disease awareness, and messaging considerations help drive an improved patient experience.
From a Whisper to a Pre-Launch Shout
Even with the most sophisticated market research techniques and technologies, the patient voice will remain unheard throughout clinical development without significant company investment in patient listening. Companies must now commit time, money, and resources as early and as enthusiastically as possible. By building trust and nurturing patient relationships, pharma benefits downstream when critical patient insights translate into successful brand decisions.
From patient journey studies to detailed feedback about unmet needs, efficacy and safety concerns, adherence considerations, reimbursement support, and advocacy, candid feedback from patients early on in the development cycle leads to improved post-launch engagement. Only by creating a culture where these insights are valued and where their acquisition is assured can pharma brands keep pace with the increasing demands of their stakeholders.
Until recently, gaining transparency into the thoughts and sentiments of patients fell exclusively into the realm of quantitative and qualitative research. Novel approaches fill the gap between the broad but shallow results of quant, and the deep but narrower revelations of qual. This gives clinical researchers agile and scalable ways to incorporate the patient voice throughout the drug development process. The result is empowered patients, improved drugs, and better healthcare.
By combining their dedication to patients with an embrace of the next generation in market research capabilities, pharma is poised to catch up to the tech firms paving the way for the ideal consumer experience. Given how healthcare is often a matter of life and death, no initiative is more significant than improving public health through the same tools we use to shop, commute, and entertain ourselves. Patients are speaking, and pharma must listen every step of the way.









