As the cost of pharmaceutical and biotechnology therapies continues to rise, concern about the ability of healthcare systems to afford high-cost therapies is accelerating. Policymakers have recently proposed initiatives to try to address the issue, including mandated price negotiations and limits on price increases. Concurrently, manufacturers highlight the high cost of clinical trials and the risks they assume in developing innovative therapies and the need to recoup the cost of their investment. Both sides of the debate beg the question: What is fair?
Our team at CRA conducted a survey of U.S. stakeholders including more than 160 physicians (rare disease specialists and oncologists) to assess their opinion on the fairness of drug pricing.1 Participants were presented with a variety of scenarios for simplified “archetype” therapies in several categories, which included:
- chronic therapies to treat rare diseases
- one-time curative therapies
- therapies to treat a rare type of cancer
The research approach draws upon a framework for pricing fairness originally introduced by Nobel Prize winning economists Daniel Kahneman and Richard Thaler, and co-author Jack Knetsch, in the pioneering paper “Fairness as a Constraint on Profit Seeking: Entitlements in the Market.”2
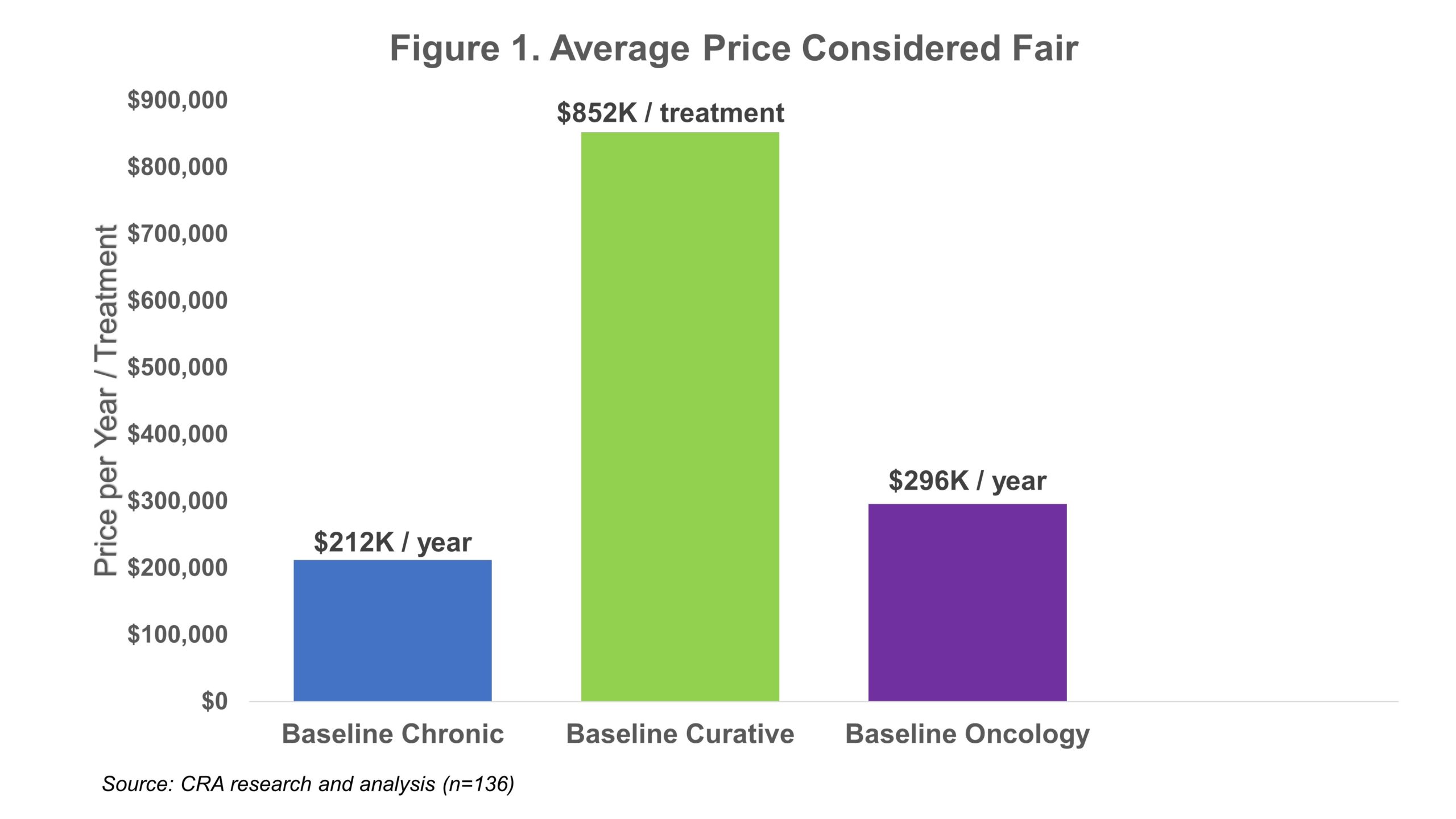
Establishing a Baseline
In the survey, physicians were asked to identify the maximum price they would consider to be fair for each of these treatment archetypes based on limited information, including the average age of the patients (50 years old), size of the patient population (1,500 U.S. patients), and other information about the target disease (severe and life-threatening). Defining these treatment archetypes as broadly as possible allowed us to evaluate a wide range of scenarios to identify the primary factors that drive perceptions of fairness. It also helped avoid confounding interpretations of results due to pre-existing views about the price or clinical performance of currently available drugs. Physicians rated the fairness of specific price levels on a four-point scale (very unfair, somewhat unfair, somewhat fair, very fair). If they responded that a price was fair (i.e., somewhat fair or fair), we asked additional questions to determine the maximum price that would be considered fair (see Figure 1).
 What Factors Impact Physicians’ Perception of Fairness?
What Factors Impact Physicians’ Perception of Fairness?
Once baseline fair prices were established, we sought to determine which factors associated with pricing of the archetype therapies were most likely to affect the perceptions of fairness, including: (a) the severity of the condition, (b) the size of the patient population, and (c) the age of the patient (adult vs. pediatric). Respondents were presented with variations of the baseline archetypes which included changes in the characteristics of the treatment and the patient population and condition, to assess their relative importance to the prices perceived to be fair (see Figure 2).
When considering different scenarios involving therapies for chronic rare conditions, most physicians (70%) agreed that the baseline price would still be considered fair if the condition was only severe but not life-threatening. On average, physicians thought that in this scenario the fair price would be 9% lower than the baseline. In contrast, for the curative archetype, 59% of physicians thought that their baseline price would still be fair and on average the physicians considered a price of 18% lower than the baseline to be fair in the “not life-threatening” scenario.
When considering a lower incidence of 200 patients for chronic rare conditions, the majority (70%) thought that a higher price would be fair and, on average, that a price 24% higher than their baseline price would be fair. This contrasts with the 40% of physicians that thought a higher incidence of 50,000 patients would warrant a lower price than their baseline and, on average, indicated that a 19% lower price would be fair. These findings are similar for the curative archetype, with 59% and 51% of physicians indicating that a higher or lower price than their baseline would be fair for a patient population of 200 and 50,000, respectively.
 Across each of the three archetypes, physicians were aligned that prices higher than their baseline price would be fair if the condition affected pediatric patients rather than adult patients. On average, in all three archetypes, the price perceived to be fair for a therapy to treat pediatric patients was more than 10% higher than the baseline fair price.
Across each of the three archetypes, physicians were aligned that prices higher than their baseline price would be fair if the condition affected pediatric patients rather than adult patients. On average, in all three archetypes, the price perceived to be fair for a therapy to treat pediatric patients was more than 10% higher than the baseline fair price.
Are Prices of Existing Therapies Fair?
In our research, we also asked respondents to provide insights regarding several therapies that are already approved and commercially available and exhibit a range of list prices and disease prevalence.3 For therapies used to treat rare chronic conditions, 44% to 58% of physicians on average indicated the prices of rare chronic disease treatments are fair, 38% to 71% believed the price of existing curative therapies to be fair, and 40% to 51% indicated that the oncology products were priced fairly.
Conclusion
Understanding the factors that physicians consider most important when assessing the fairness of drug prices can play an important role in developing pricing and market access strategies for new and innovative therapies. Physicians agreed that higher prices are fairer for therapies indicated for smaller patient populations or pediatric patients. In contrast, lower prices are considered fairer for therapies indicated for severe but not life-threatening conditions or therapies approved to treat significantly larger patient populations. Moving forward, discussions among stakeholder groups must continue to try to align on what is “fair” for pricing of different drugs, especially as more drug developers invest in rare disease therapies, including potentially curative treatments, and work to make these drugs broadly available to patients in need.
References:
1. The research was sponsored by several pharmaceutical and biotechnology manufacturers.
2. Kahneman, Knetsch, Thaler, “Fairness as a Constraint on Profit Seeking: Entitlements in the Market,” American Economic Review (September 1986, Vol. 76, No. 4).
3. WAC (wholesale acquisition cost) prices for competitor products were specified as the reference price.
The authors wish to acknowledge the contributions of Dr. Kristen Backor and Joseph Black to this article.
The views expressed in this paper are those of the authors and do not represent the views of Charles River Associates.








