Understanding and bettering the patient journey is essential to delivering on the promise of the specialty pipeline to improve outcomes and increase efficiency in healthcare. From a patient’s ability to afford medication to a payer’s coverage of products that provide advances in treatment, each step of the healthcare chain is important to ensuring a patient can benefit from the results of today’s specialty drugs.
Increasingly, orphan drugs, which often fall under the specialty designation, are becoming a focus area for payers because pressures to manage costs are continuing to rise and treatments for some therapeutic areas are becoming more common. The Orphan Drug Act (ODA) encourages the development of treatments by providing financial incentives to manufacturers. The ODA offers a tax credit on R&D costs, R&D grants for Phase I to Phase III clinical trials, and seven years of marketing exclusivity from approval.
As manufacturers see opportunities to help patients by developing rare diseases therapies along with the added incentives of the ODA, payers are increasingly monitoring expenditures related to specialty therapies in the orphan drug space. Payer business models are shifting in anticipation of growing patient populations and costs, and increased management of rare disease/orphan drugs is likely to increase by 2015-2016.
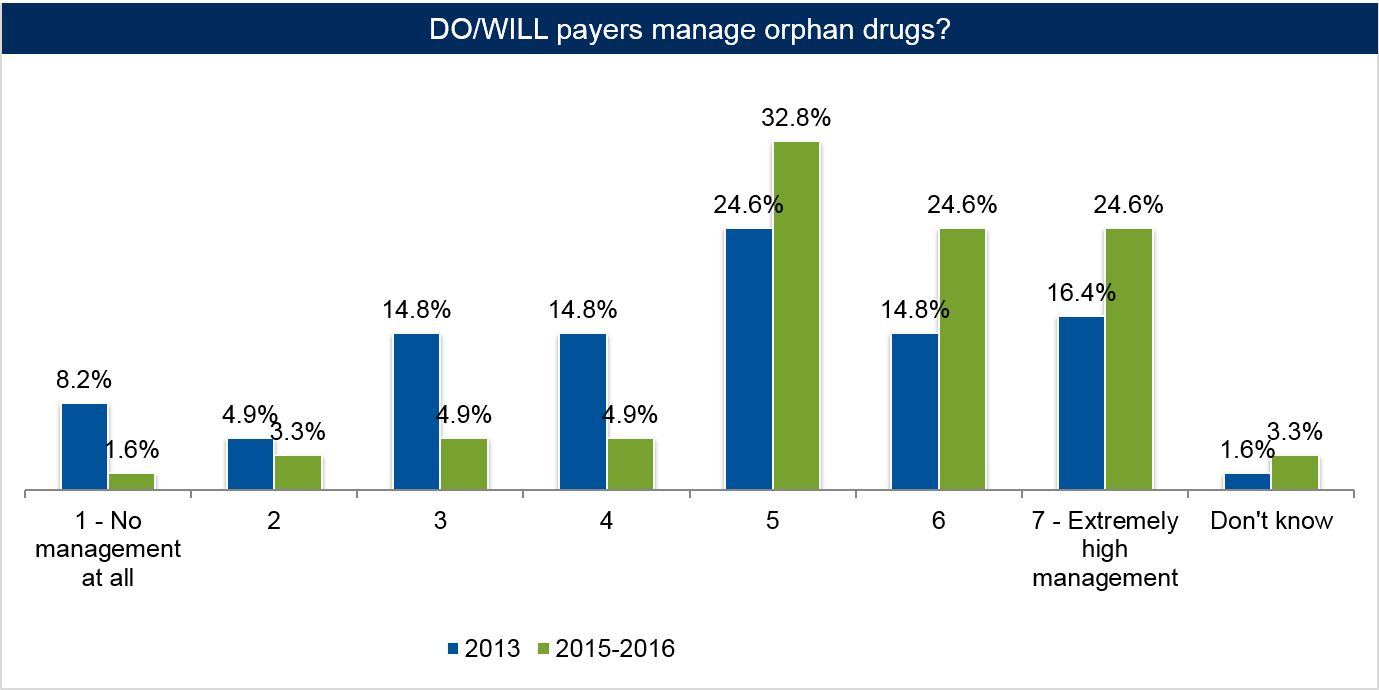
Key Questions to Address to Improve Access
Manufacturers must align the speed of the R&D process with a deep understanding of the patient journey to make key access decisions along the way. The following questions highlight important considerations for facilitating access and adherence once the product is available to patients:
- Are there payer or provider education needs around the disease state or type of treatment?
- How can the treatment be demonstrated to provide a better outcome for patients and a more efficient result for payers?
- What method of distribution will best support patients in starting treatment early and staying on treatment while ensuring safe use?
- How will pharmacies be a partner in access and adherence?
- What patient support programs can be offered for the unique access and adherence challenges?
- Are reimbursement or co-pay support services necessary?
- How many patients may be uninsured and in need of access to medication for free?
- Will patients need nurse support or training on self-administration of therapy or other management of their condition?






