FROM CANCER EPIDEMIOLOGY
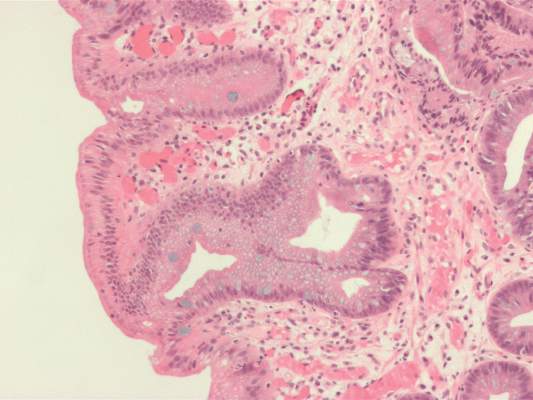
Australian researchers who analyzed 233 esophageal adenocarcinoma tumor specimens found no evidence of human papillomavirus DNA.
The findings run counter to those from a smaller, separate study from Australia led by Dr. Shanmugarajah Rajendra, in which HPV was detected in 67% of esophageal adenocarcinoma tumor specimens ( Am. J. Gastroenterol. 2013;108:1082-93).
Although some researchers have speculated that HPV might be involved in cancers of the upper aerodigestive tract, “the concept is controversial,” authors of the current analysis, who were led by Dr. Annika Antonsson , wrote in an article published online Feb. 16, 2016 in Cancer Epidemiology. “In support of this hypothesis are the observations that HPV DNA is increasingly found in cancers of the oropharynx, in particular tonsillar SCCs [squamous cell carcinomas], and HPV has recently been reported at high frequency in BE [Barrett’s esophagus] and EAC [esophageal adenocarcinoma] tissues.”
Dr. Antonsson of the department of population health at QIMR Berghofer Medical Research Institute in Brisbane, Australia, and her associates set out to determine the prevalence of HPV DNA in 233 Australian patients with adenocarcinomas of the esophagus and gastro-esophageal junction ( Cancer Epidemiology. 2016 Feb 16. doi: 10.1016/j.canep.2016.01.014 ). The mean age of patients was 64 years, 89% were male, and 79% were overweight or obese.
The researchers used polymerase chain reactions (PCRs) to test for DNA quality and for the presence of HPV DNA. Each sample was tested three times, but none of the 233 samples tested positive for HPV DNA.
Dr. Antonsson and her associates noted that the discordance with findings from the smaller Australian study from 2013 could be explained by differences in approach and design. For example, the smaller study involved patients from just two hospitals, while the current analysis involved patients from hospitals in all states of mainland Australia. Also, “we used FFPE tumor specimens for analysis, unlike Rajendra et al. who tested fresh tissue samples with a nested PCR using MY09/11 primers in the first PCR reaction, and then GP5+/GP6+ primers in the second PCR reaction,” the researchers wrote. “Since the MY09/11 PCR product is 450 bp, and we used FFPE tissue [fragmented tissue and primer pair should yield a PCR product 200 bp or shorter], we used a single-run GP + PCR approach with our samples.”
The study was funded by a grant from the National Health and Medical Research Council of Australia. The researchers reported having no financial disclosures.