Med Device: NASA Develops New Medical Device
Space is the final frontier for a new hand-held, super light device, used to diagnose medical conditions during flight. A team of six scientists from NASA and Lawrence Livermore National Laboratory (LLNL) are currently testing the device, which gives astronauts the ability to evaluate their health periodically to monitor the long-term effects of space travel, including bone degeneration, heart and eye disease, and diseases from exposure to microgravity and ionizing radiation, which are suspected but not well understood.
According to Matt Coleman, a radiobiologist from LLNL, “Some of the tools we’re developing will be among the first multi-function health diagnostic devices used in space.” The new device detects virtually any biomarkers or molecules that can indicate radiation exposure, from the blood, saliva, and breath.
Doctor Docs: An App Detects Premature Birth Risk
Doctors can now tell if a pregnant woman is at risk for a premature birth through an app developed at King’s College London. After a physician inputs important information about previous births, cervix length, and levels of fetal fibronectin, the app uses an algorithm to determine the risk. Two studies published in Ultrasound in Obstetrics & Gynecology journal proved that the app worked much better as a predictive tool than a physician would considering these factors alone.
Of the 15 million babies born prematurely worldwide, one million die from complications. The scientists at King’s College London believe that better prediction of premature birth will allow for better care of the mother throughout pregnancy and a more successful birth with proper preparation.
Sales Sector: Treato Ranks Popular Statins
Merck has the number one brand name and generic statins, according to Treato, a consumer healthcare website which ranked statins by their price and user satisfaction. Merck’s Vyotin received 2.8 out of 5 stars when users were asked to indicate their satisfaction with the drug. The company’s generic version, lovastatin, got a 2.7.
While users rated their satisfaction based on the positive and negative side effects, Treato also includes the price of each statin. Vytorin came in at $233.53 for a 20 mg, 30-day supply and lovastatin at $4.69 for a month supply. Bristol-Myers Squibb’s Pravachol had the most negative concerns discussed, while lovastatin had the least.
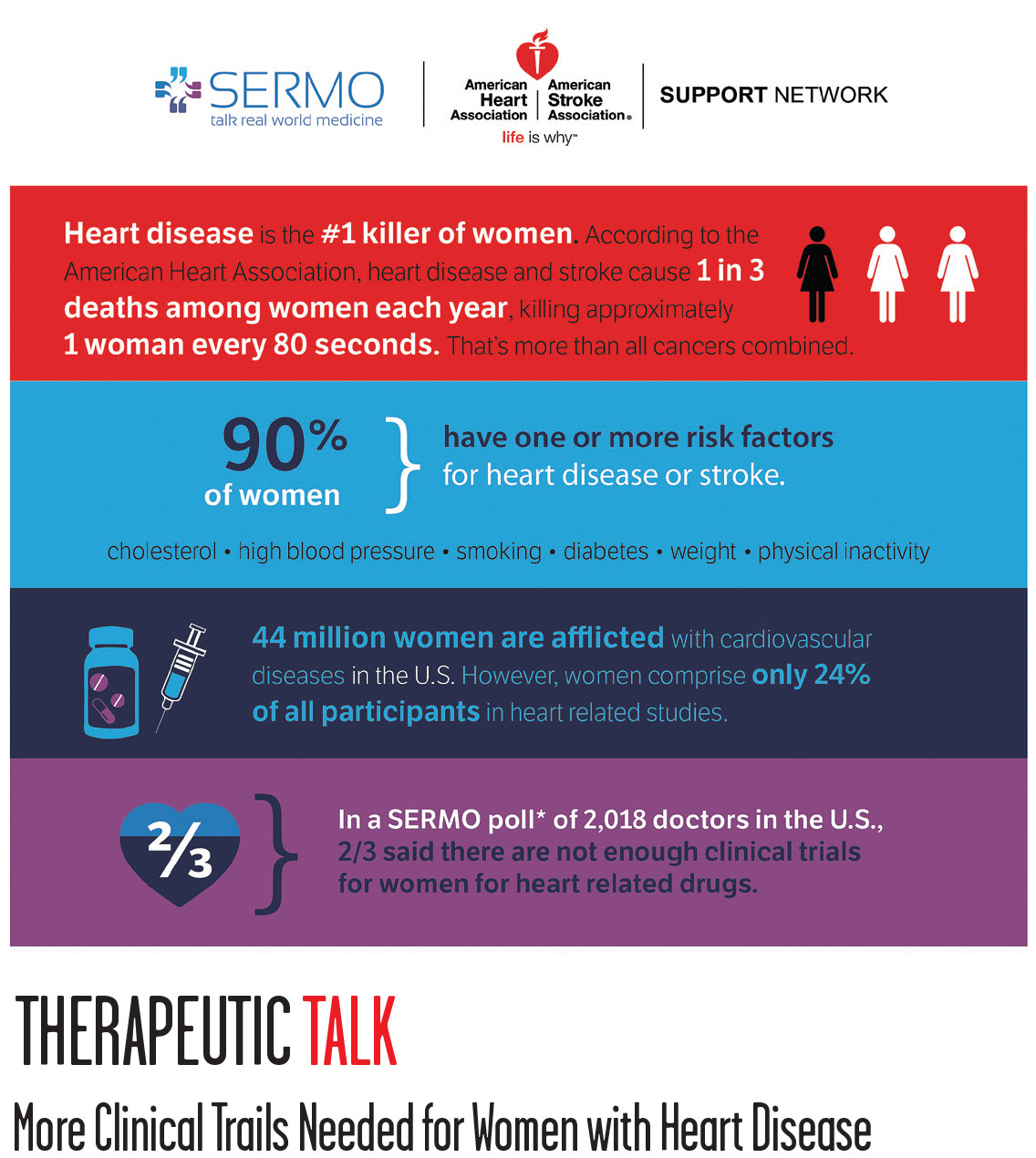
Therapeutic Talk: More Clinical Trails Needed for Women with Heart Disease
Heart disease remains the number one cause of death for American women, but the reason is not due to bad lifestyle habits or genetics. Among 2,000 physicians recently surveyed by SERMO in conjunction with the American Heart Association, more than two-thirds of physicians say that there are just not enough clinical trials for women. At the same time, in the clinical studies that are conducted, only 24% of the 44 million women in the U.S. with cardiovascular diseases actually participate. The stats lead to the question: Why have women been left out of heart disease studies and why is it so important to include them?
“For years, we were taught that cardiovascular disease was more of a male disease,” says Dr. Linda Girgis, a New Jersey-based physician. “Risk factors were not so well stratified in women as in men. Within the last several years, we are realizing that cardiovascular disease is also a leading killer of women and they have long been neglected in heart disease studies.”
Discoveries/Innovations: Surprisingly Simple Ways to Stop Sting Deaths
Despite the fact that box jellyfish stings kill more people than shark attacks annually, doctors have not yet found the best way to treat them. Researchers at the University of Hawaii at Manoa have come up with a new method to safely test all treatment options for stings, from the mythical urine treatment to military grade medicine. Lead author of the study, Angel Yanagihara, comments, “We designed a set of experiments using live, stinging tentacles and live human red blood cells which allowed us to pit first-aid measures against one another.”
Yanagihara and his team found surprisingly simple methods are the most effective at treating these stings. Submerging the sting in hot water and vinegar is almost as effective as Sting No More, a venom inhibitor developed with Department of Defense funds.
FDA Update
Free Biosimilars Course for HCPs
A 90-minute online course on biosimilars for HCPs, titled the FDA Overview of Biosimilar Products, was recently released on the FDA website. The course offers important information about the production, use, and functions of approved biosimilars.
The regulatory group created the continuing education program to help physicians make more informed decisions when considering and prescribing biosimilars.
Drug Approvals
Eisai’s Halaven (eribulin mesylate), received FDA second-indication approval as an injectable treatment (0.5 mg) for those with unresectable or metastatic liposarcoma who have already received chemotherapy. The orphan drug is the first single agent to demonstrate a significant extension of the survival rate in patients at this stage of the disease.
Orphan Drug Designation
BioMarin received orphan drug designation for the first AAV-Factor VIII Gene Therapy, BMN 270, for patients with Hemophilia A, a genetic disorder caused by a missing or defective factor VIII, a protein essential for blood clotting. The company will report on its Phase 1/2 study in April.
Med Device Approvals
The FDA has approved a new model of Olympus’ TJF-Q180V duodenoscope, used as a minimally invasive way to drain fluid built up in pancreas and bile ducts blocked by cancerous tumors, gallstones, or other gastrointestinal conditions. The new model sports a tighter seal, greatly reducing the risk of bacterial infection, a common side effect of the procedure.