Psoriasis and related conditions represent as an evolving treatment domain for many pharmas—and obvious categories of growth for those with existing biologic and JAK inhibitor franchises. New data from U.S. dermatologists show that they want more oral treatments with improved clinical profiles in 2019—and more affordable options.
 With recent research demonstrating that moderate to severe psoriasis carries increased risk for cardiovascular disease, psoriatic arthritis, obesity, and depression, in addition to already compromised quality of life, pharma innovation in this domain can deliver a deeply human benefit.
With recent research demonstrating that moderate to severe psoriasis carries increased risk for cardiovascular disease, psoriatic arthritis, obesity, and depression, in addition to already compromised quality of life, pharma innovation in this domain can deliver a deeply human benefit.
Data sourced by InCrowd, a real-time market insights technology firm serving the life sciences, show:
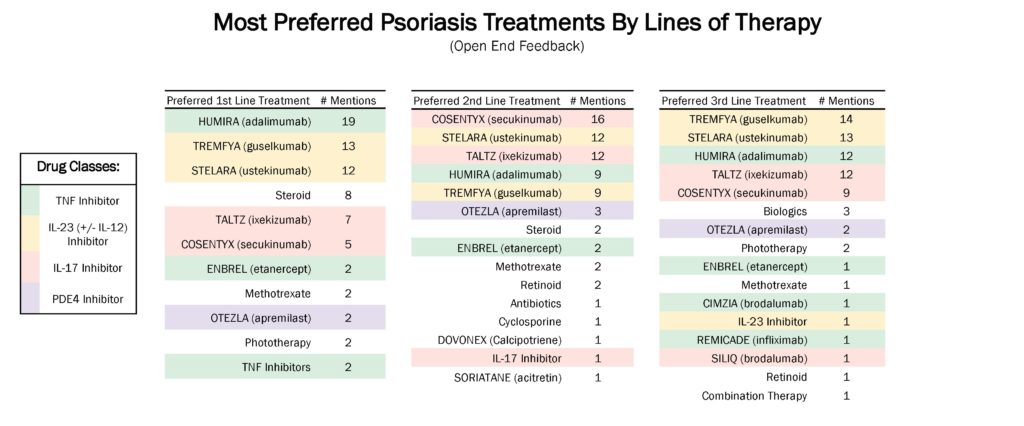
- Almost half of moderate to severe psoriasis patients are being treated with biologic therapies, according to the data. Humira and Stelara are the most commonly prescribed biologics for moderate to severe psoriasis. In indicating top-of-mind preferences for treatments, physicians highlighted Humira, Cosentyx, and Tremfya as first-line, second-line, and third-line treatments respectively. Of note, Stelara is mentioned among the top three choices across all lines of therapy.
- Despite strong biologics treatment prescribing patterns, the data show that corticosteroids were the #1 prescribed treatment among respondents, at 58%, compared to biologics at 48%.
- Treaters of psoriasis are excited about new therapeutic options available to them, highlighting IL-17/23/31 agents, JAK inhibitors, and TYK2 inhibitors, also noting interest in more orally administered therapies. While some respondents weren’t excited about any of the new options, more than 10% proactively named Risankizumab when asked about exciting new treatment options. “I’m not excited about any (new treatment options) because they seem like ‘me too’s,’ but I hear Risankizumab is going to be good,” said one responding dermatologist from Texas with seven years in practice.
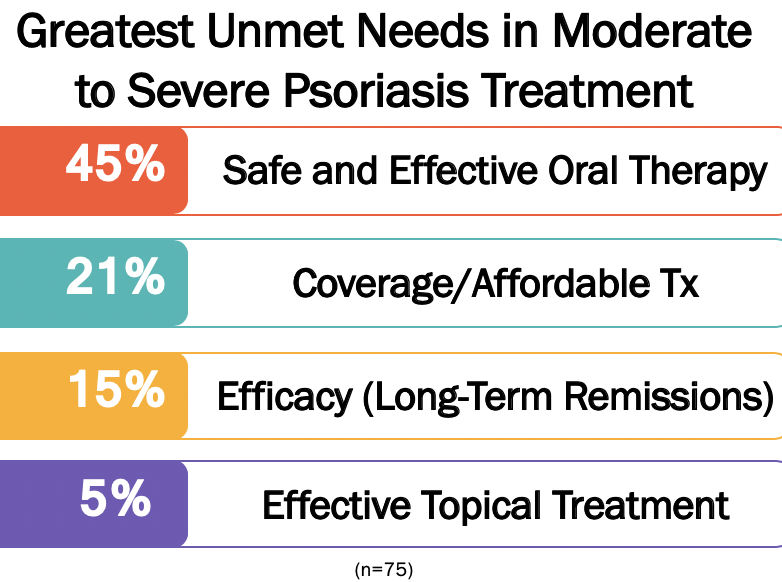
- Safe, effective, and affordable oral treatments are the greatest unmet need in the treatment of moderate to severe psoriasis—as cited by 45% of respondents.
- Increased affordability for patients, and treatments that achieve long-lasting remission were other unmet needs that ranked second (21%) and third (15%) respectively. “It doesn’t matter how good a medicine is if the patient can’t afford it,” said one dermatologist from Connecticut with 14 years in practice.
 Verbatim remarks offer a view into real-world prescribing considerations. “Even with insurance, out of pocket costs are too high and prior authorization process is too burdensome and takes too long,” said a dermatologist practicing in Washington state.
Verbatim remarks offer a view into real-world prescribing considerations. “Even with insurance, out of pocket costs are too high and prior authorization process is too burdensome and takes too long,” said a dermatologist practicing in Washington state.
Numerous respondents said that many patients hate needles and thus find injectable treatments uncomfortable. “We need the high efficacy of the injectable biologics in the form of an oral agent,” said a dermatologist from Ohio with 22 years in practice.
One respondent noted that comorbidities such as cancer often preclude the use of immunosuppressive agents.
As pharmas define their agendas for 2019, the voice of the clinician in the form of this new data can help inform pharma marketers as their brands expand in the space.
InCrowd’s psoriasis treatments perceptions survey included 75 responding dermatologists who participated in a five-minute microsurvey between Nov 21-26, 2018. Each respondent has treated at least 20 patients with moderate to severe psoriasis in the past three months—with the average of 79 patients. Each characterized themselves as moderately or more aware of products in development for the disease.