Curavit Clinical Research
Dave Hanaman, President and Chief Commercial Officer
dave.hanaman@curavitclinicalresearch.com
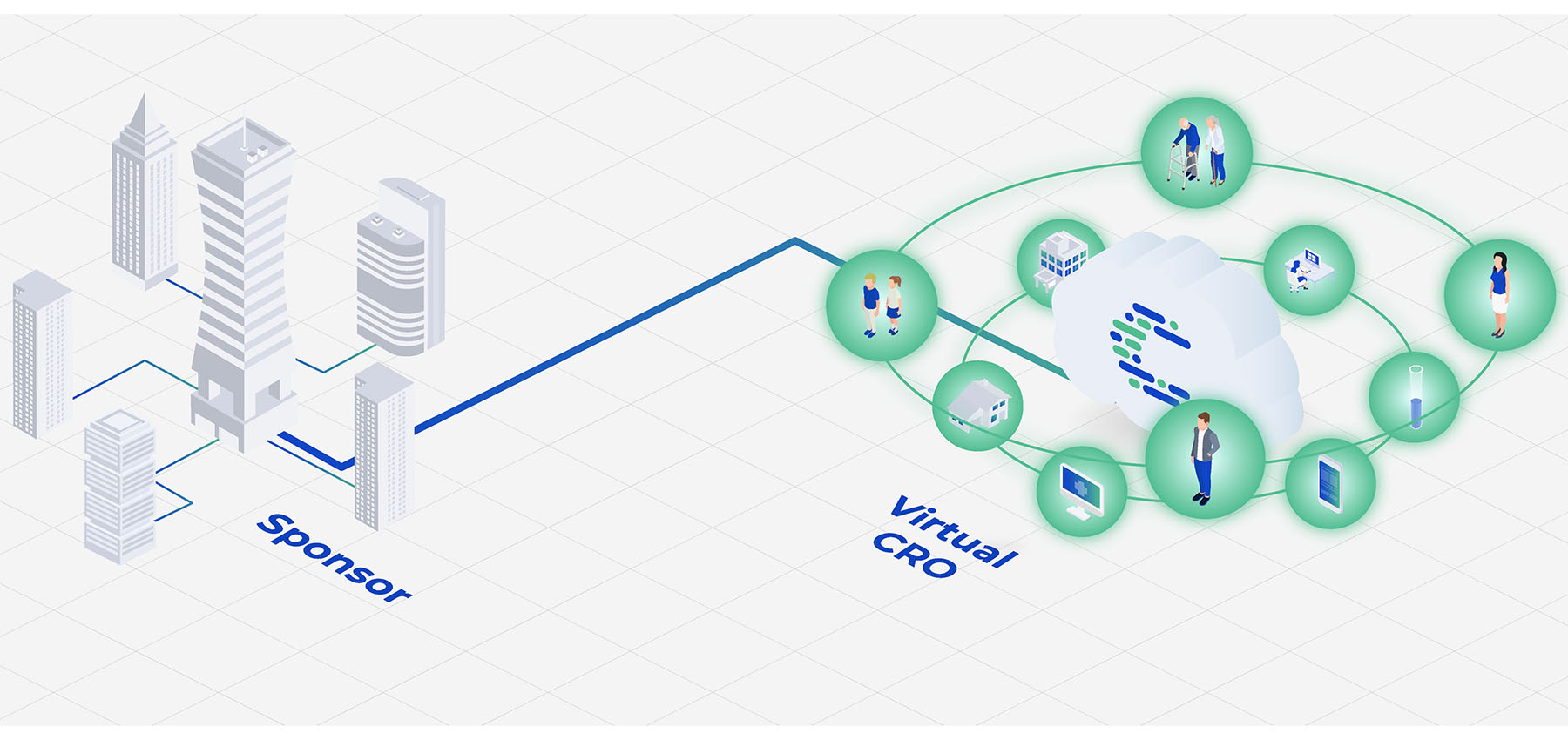
Launched in December 2019, Curavit Clinical Research is disrupting traditional clinical trials as the first virtual contract research organization (VCRO). The company applies digital-first principles to design and execute decentralized clinical trials (DCTs), leveraging its proprietary central platform, built on proven, industry-leading technology and integrating with all leading DCT software.
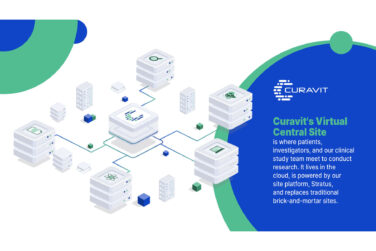
Curavit’s platform is the always audit-ready digital manifestation of its industry leading experience and proven processes. It is uniquely configured for each trial to manage every aspect, ranging from operations, to data collection and insight, to patient experience. Using the platform, Curavit’s expert clinical operations team interfaces in real time, in the cloud with patients, partners, and sponsors to conduct world-class research.
For example, Curavit partnered with Beech Tree Labs in September 2020 to design and execute a fully virtual clinical trial that was ready in just eight weeks to investigate a promising new COVID-19 therapy. The clinical study evaluated the effect of the company’s therapy on mean duration and severity of disease in outpatients aged 60 years or older with underlying conditions. This fully virtual clinical trial was made possible by applying Curavit’s digital-first principles design philosophy to employ telehealth, electronic patient-reported outcomes (ePRO), and full web and contact center patient support to eliminate the need for in-person office visits.
Curavit’s digital-first design results in decentralized, hybrid, or compound trials that solve a very specific challenge, such as endpoint adjudication to centralize and ensure quality across patients, sites, and time. By minimizing the need for physical infrastructure and “bringing the trial to the patient,” Curavit is leading the way to democratize patient recruitment, improve data quality, increase trial speed, and reduce costs.