Trial Landscape
H1
Rob Consalvo, Senior Director of Sales Engineering & Enablement
robert.consalvo@h1.co
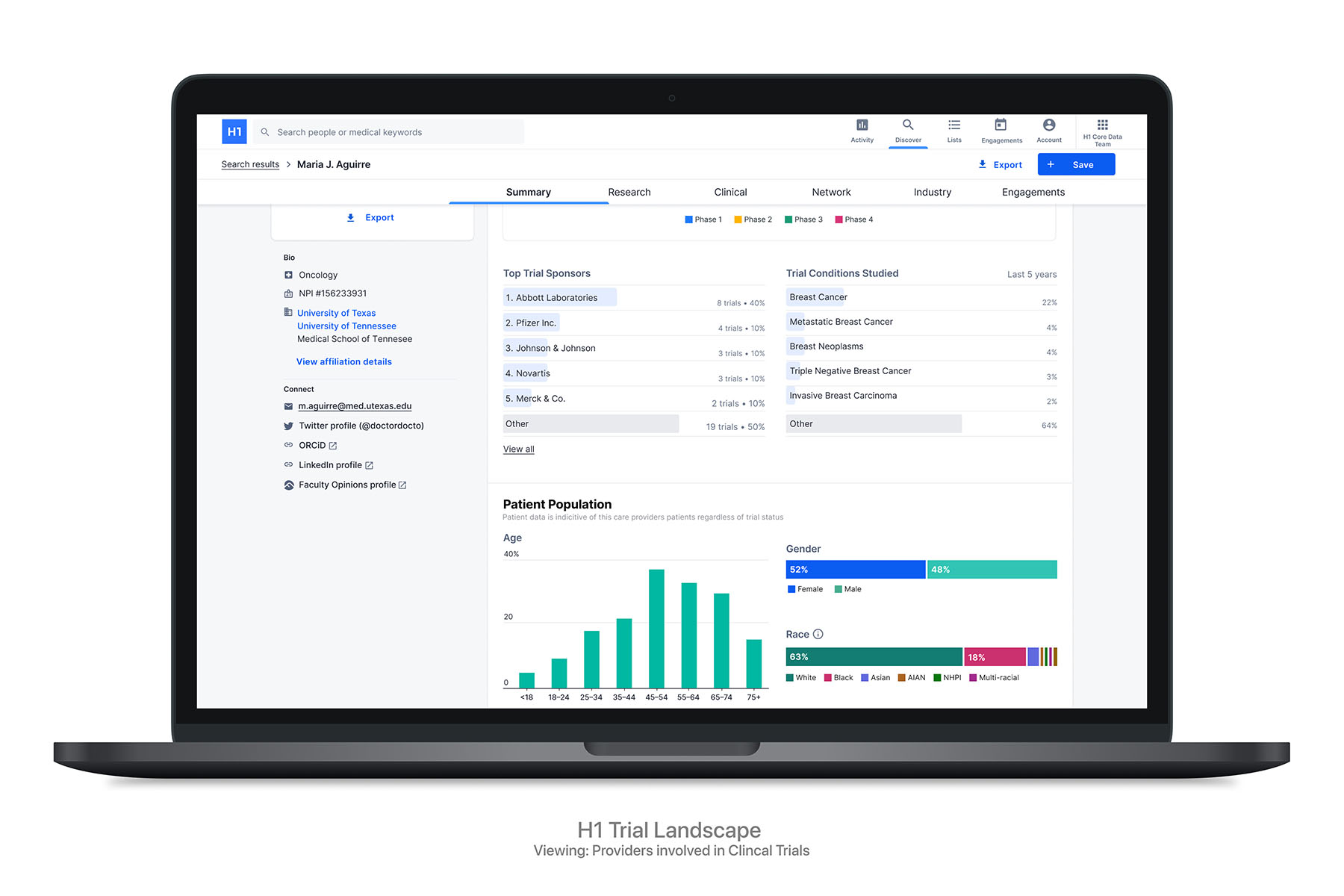
More than 80% of clinical trials fail to enroll patients in a timely manner, causing delays in the trial process, according to a study published in Perspectives in Clinical Research. To help solve this, H1 launched Trial Landscape in September 2021. This solution can allow pharma and life sciences companies to determine the required clinical experience levels for their clinical trials; analyze patient segments to help improve inclusivity, retention, and recruitment; better understand locations and investigator profiles for their specific trial type globally; and complete feasibility much more quickly, often in hours instead of weeks.
Trial Landscape is already in use at four companies, three of which are top 10 pharma companies. One client was struggling to find new potential investigators and sites in specific regions who had the right experience levels in immunology and infectious disease, as well as have access to diverse patient populations for an upcoming vaccine clinical trial. The company had already exhausted its internal resources but did not have any information to support the new diversity requirements the FDA made for clinical trials in 2020.
By leveraging a number of different sources such as claims data, H1 helped provide information about the segmentation and diversity of patients being treated by different providers and sites. The H1 team also used a number of provider diversity proxies such as languages spoken by the provider or self-identified race, ethnicity, and gender to help better understand how providers and sites relate and identify with different patient populations. The client anticipates that the H1 Diversity and Inclusion data will provide them with a market analysis resulting in 60% increase in profiled people and institutions.