PixCell Medical
Hanan Ben-Asher, COO and Business Development
hanan@pixcell-medical.com
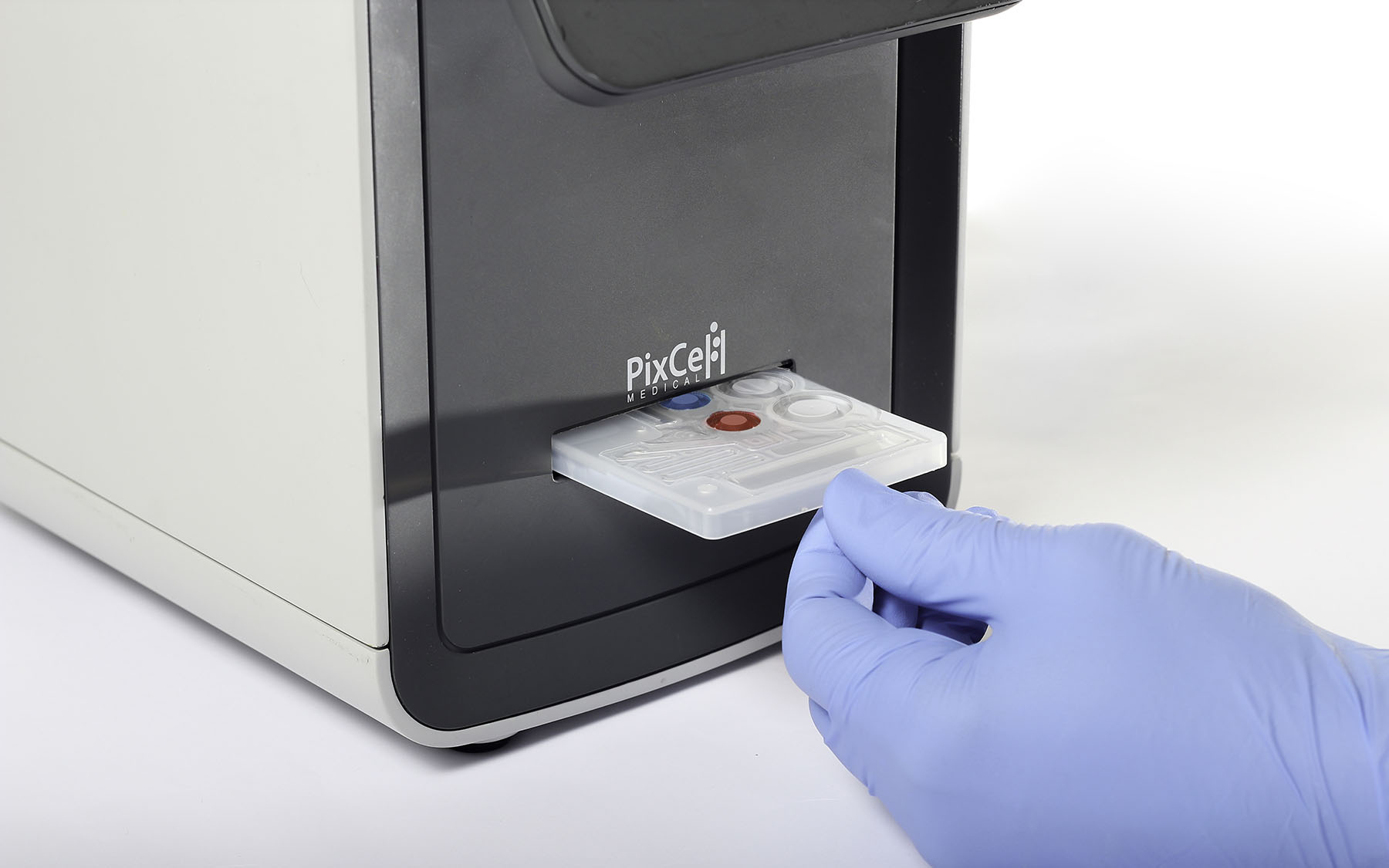 PixCell Medical has developed the only FDA-cleared platform intended for use at the point of care that can deliver lab-quality, full five-part differential Complete Blood Count (CBC) results. Leveraging the company’s patented Viscoelastic Focusing technology, along with AI-powered machine vision, PixCell’s HemoScreen diagnostic platform shortens diagnostic results delivery from days to minutes. With just one drop of blood and within six minutes, the HemoScreen delivers accurate readings of 20 standard blood count parameters, saving patients, clinicians, and health systems significant time and costs. A study performed in Sweden demonstrated that turnaround time for CBC results at the ER was reduced by 30 minutes when using the HemoScreen.
PixCell Medical has developed the only FDA-cleared platform intended for use at the point of care that can deliver lab-quality, full five-part differential Complete Blood Count (CBC) results. Leveraging the company’s patented Viscoelastic Focusing technology, along with AI-powered machine vision, PixCell’s HemoScreen diagnostic platform shortens diagnostic results delivery from days to minutes. With just one drop of blood and within six minutes, the HemoScreen delivers accurate readings of 20 standard blood count parameters, saving patients, clinicians, and health systems significant time and costs. A study performed in Sweden demonstrated that turnaround time for CBC results at the ER was reduced by 30 minutes when using the HemoScreen.
With nearly a decade invested in developing this novel technology, the HemoScreen was cleared by the FDA in December 2018. Since the release of the HemoScreen in 2019, it has been deployed in numerous countries, serving clinicians in settings where speed and accuracy of results are critical, including operating rooms, oncology wards, and emergency centers. The HemoScreen is portable, requires no installation nor any advanced technical training for use.
The HemoScreen has also recently proven to be an important tool in the management and monitoring of patients diagnosed with COVID-19 in Uppsala University Hospital in Sweden. Additionally, over the course of the year, PixCell has provided 50 HemoScreen devices to the New South Wales Ministry of Health for use in small labs, emergency departments, and remote communities without widespread access to healthcare, making important tests more accessible. The HemoScreen has been successfully evaluated in several top-tier university hospitals in the United States and is expected to launch in the country in 2021.