Novocure
Jaclyn Stahl, Senior Manager – Brand Public Relations
jstahl@novocure.com
 Novocure is a global oncology company with two FDA-approved products: Optune for the treatment of glioblastoma and Optune Lua for the treatment of mesothelioma. But these treatments are innovative for another reason—they are a portable and wearable device that tracks data specific to each patient’s usage. To ensure treatment is effective, the company offers an award-winning patient service called nCompass where a device support specialist (DSS) provides treatment education and initiation service in person at either the patient’s home or at the healthcare provider’s office. Typically, the DSS would also visit each patient monthly to download data on how much the device was used, or the device itself would need to be shipped back to Novocure to extract the data.
Novocure is a global oncology company with two FDA-approved products: Optune for the treatment of glioblastoma and Optune Lua for the treatment of mesothelioma. But these treatments are innovative for another reason—they are a portable and wearable device that tracks data specific to each patient’s usage. To ensure treatment is effective, the company offers an award-winning patient service called nCompass where a device support specialist (DSS) provides treatment education and initiation service in person at either the patient’s home or at the healthcare provider’s office. Typically, the DSS would also visit each patient monthly to download data on how much the device was used, or the device itself would need to be shipped back to Novocure to extract the data.
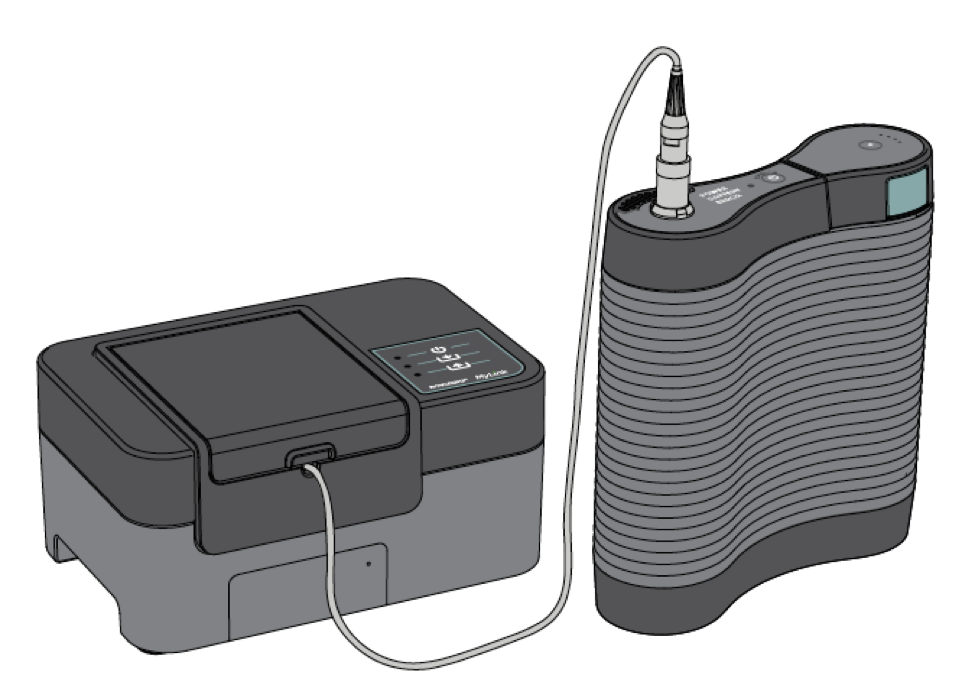
To make this process easier, Novocure had already been at work on MyLink, a new offering that would allow patients to download their data from the safety of their home, while still allowing patients the opportunity to speak live with a DSS for details about their data. Additionally, the MyLink technology also provides real-time feedback to Novocure’s trained support center to troubleshoot any issues more accurately.
But when COVID-19 hit, the completion of MyLink became a high priority. A MyLink Task Force team was created to develop strategies for immediate implementation including key performance indicators and timelines. This initiative began in April, but by May Novocure had implemented a phased launch with an initial wave of 30 U.S. patients, and a second wave of 70 patients using this technology as of June. This strategy increased Novocure’s virtual patient support capability and put information into the hands of patients without the need for in-person contact.