LightDeck Diagnostics
Kathryn Ryan, PR representative
pr@lightdeckdx.com
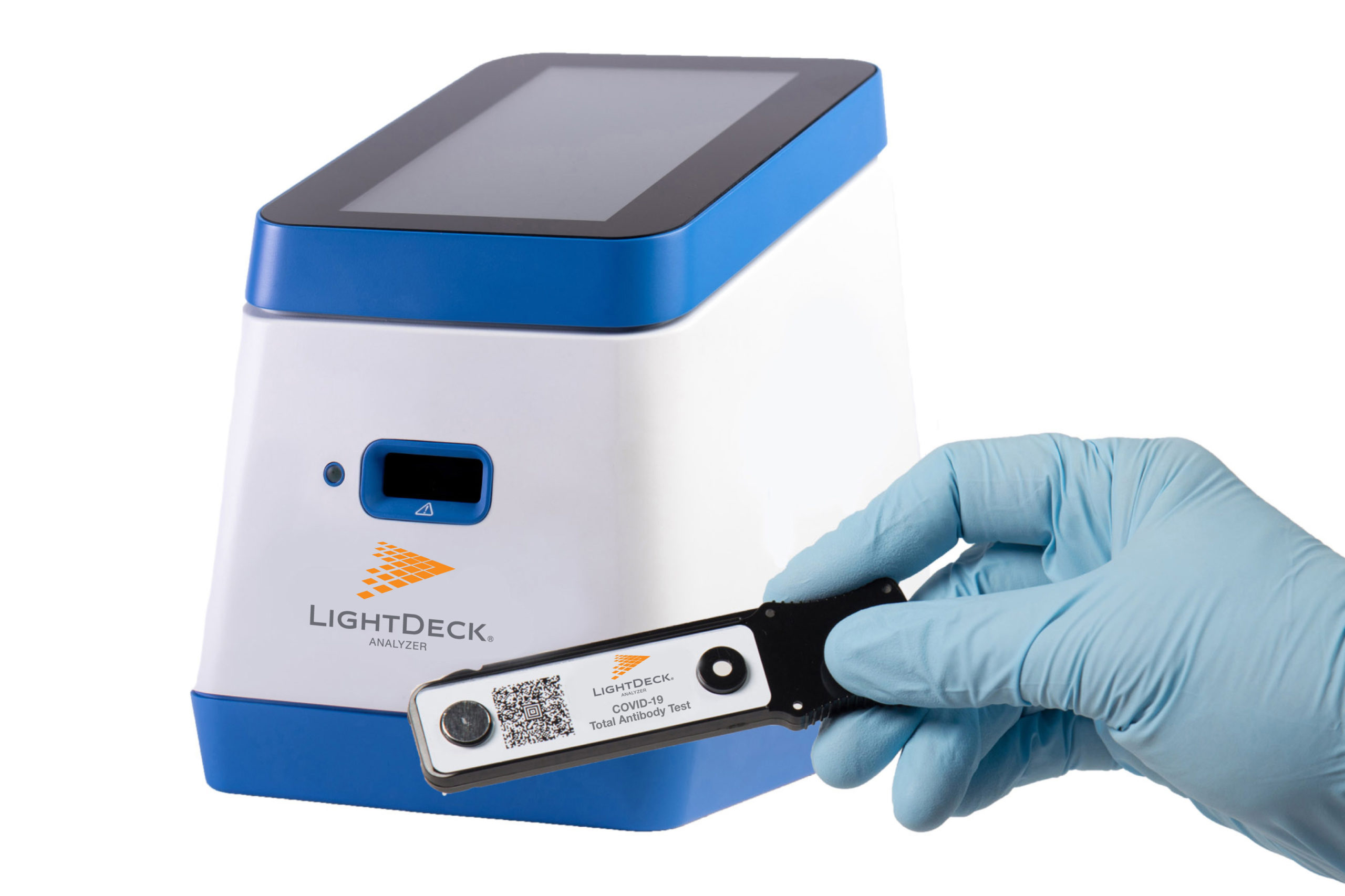 LightDeck Diagnostics is dedicated to a new healthcare approach, where quick and accurate diagnostic tests will run wherever and whenever they’re needed. The company’s proprietary LightDeck platform combines advanced planar waveguide with novel materials and patented manufacturing techniques to deliver lab-quality results anywhere, in minutes. Furthermore, the test procedure is simple enough that even an untrained user can generate accurate results, which means it can be used in emergency departments as well as schools, airports, prisons, nursing homes, and more. This will prove especially useful as the company is introducing COVID-19 tests for acute infection and past infection.
LightDeck Diagnostics is dedicated to a new healthcare approach, where quick and accurate diagnostic tests will run wherever and whenever they’re needed. The company’s proprietary LightDeck platform combines advanced planar waveguide with novel materials and patented manufacturing techniques to deliver lab-quality results anywhere, in minutes. Furthermore, the test procedure is simple enough that even an untrained user can generate accurate results, which means it can be used in emergency departments as well as schools, airports, prisons, nursing homes, and more. This will prove especially useful as the company is introducing COVID-19 tests for acute infection and past infection.
The COVID-19 pandemic has laid bare the lack of access to quick, affordable, lab-quality testing. Current antigen tests are much faster and cheaper than the polymerase chain reaction (PCR), but antigen tests compromise performance for speed. In addition to antigen or PCR tests to diagnose active infections, high-quality, point-of-care antibody tests are needed to determine a person’s immune status. LightDeck Diagnostics will market both tests and deliver lab-quality performance in just five minutes.
The company is also introducing host response panels for the diagnosis and management of acute infections and sepsis. Rather than culturing blood for 24-36 hours to identify bacteria, host response proteins are present early in the process, which enables assessment of the patient’s immune system and earlier diagnosis of sepsis. Additionally, the company is also working on cardiac panels to diagnose a heart attack or to discharge low-risk patients safely. Currently, central lab troponin tests used to detect a potential heart attack suffer from a turnaround time of 60-90 minutes, but LightDeck Diagnostics’ tests can deliver results in 15 minutes in the emergency department.








