Echosens
Katya Grudkovska, Marketing Director, North America
katya.grudkovska@echosens.com
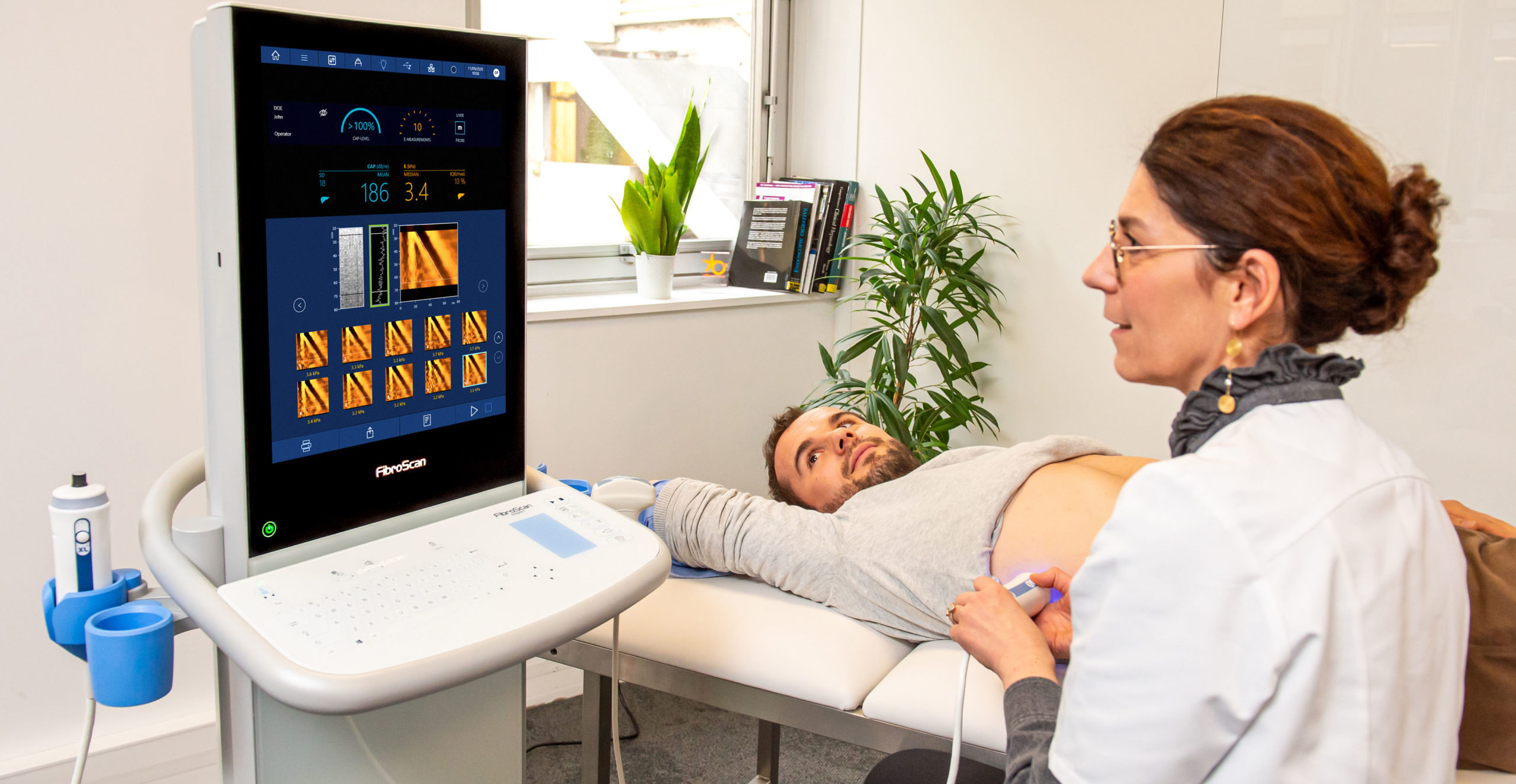
 Echosens is a high-technology company and one of the world’s top providers of non-invasive medical devices dedicated to assessment of chronic liver diseases. The company pioneered the field of elastography technology by developing FibroScan, the first device that quantifies liver stiffness non-invasively and painlessly at the point of care. FibroScan uses a patented technique called VCTE in which a controlled frequency shear wave is mechanically induced to the liver. The propagation speed of this shear wave is then measured with pulse-echo ultrasound, and a high shear wave speed correlates to high liver stiffness.
Echosens is a high-technology company and one of the world’s top providers of non-invasive medical devices dedicated to assessment of chronic liver diseases. The company pioneered the field of elastography technology by developing FibroScan, the first device that quantifies liver stiffness non-invasively and painlessly at the point of care. FibroScan uses a patented technique called VCTE in which a controlled frequency shear wave is mechanically induced to the liver. The propagation speed of this shear wave is then measured with pulse-echo ultrasound, and a high shear wave speed correlates to high liver stiffness.
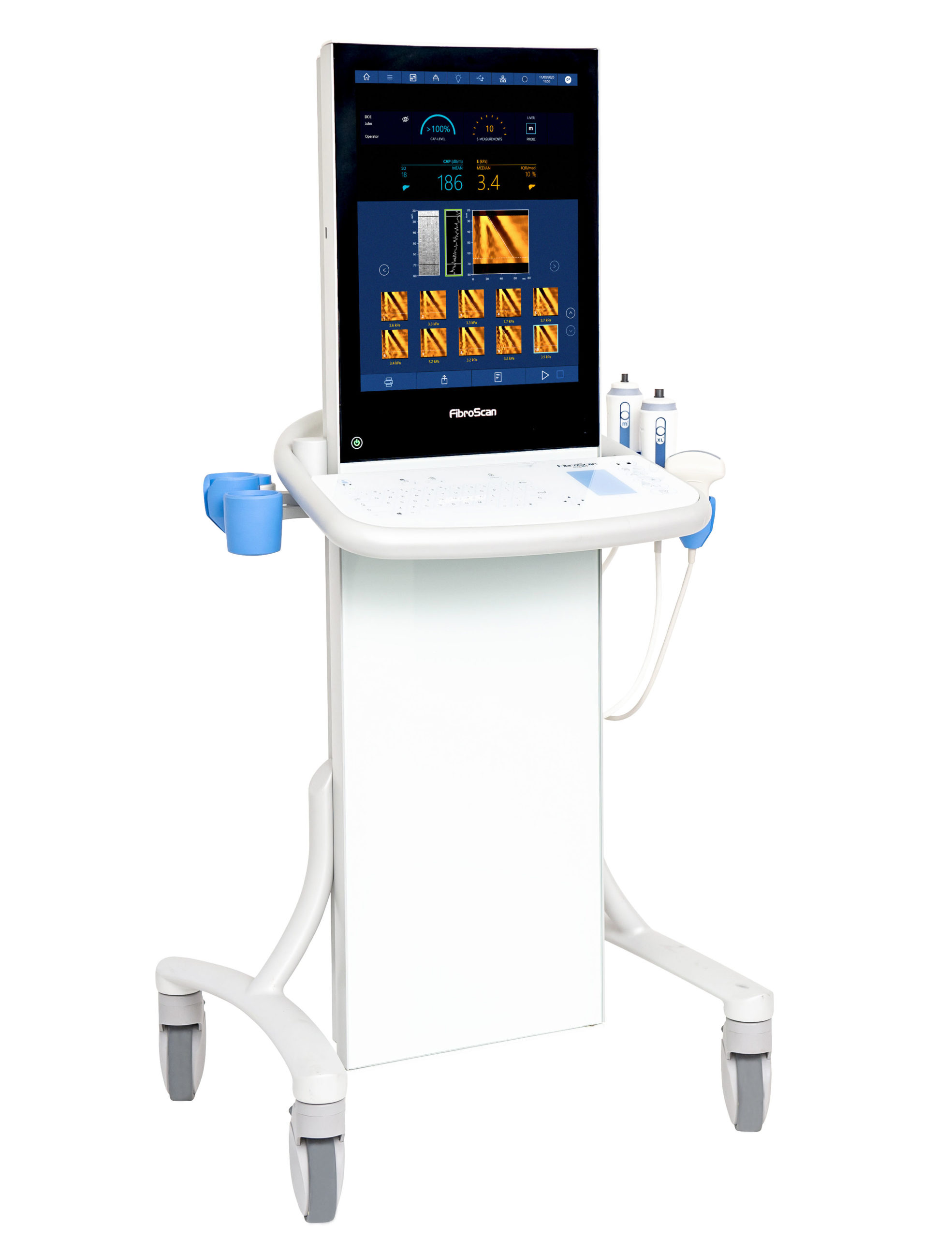
Most recently, FibroScan 630 Expert was introduced internationally in March 2020 and the U.S. in November 2020. This latest addition to the FibroScan family of products adds the capability for spleen stiffness measurement. Non-invasive assessment of the spleen is an important advancement in assessing portal hypertension (PH), which is the main consequence of cirrhosis and responsible for a majority of its complications. The American Association for the Study of Liver Disease (AASLD) guidelines recommend that all people with cirrhosis be evaluated for clinically significant PH.
The current standard for assessing PH is hepatic venous pressure gradient (HVPG), a highly invasive procedure. The spleen stiffness measurement on the FibroScan 630 Expert may provide a cost-effective and non-invasive way to assess the severity and prognosis of advanced liver disease and a comprehensive solution to manage patients with chronic liver diseases, including PH.
Additionally, FibroScan 630 Expert offers a new intuitive user interface, high-speed processing, integrated barcode reader, ultrasound localization probe (time-saving technology for locating the spleen and liver in obese and complex patients), redesigned ergonomics, touchscreen, and washable touch keyboard.