ClinicalBid
Ken Steinberg, CEO
ken@clinicalbid.com
 Launching most clinical trials is hit-or-miss. Prices are all over the map. Quality is suspect. Staffing is a nightmare and patient populations are unpredictable. What if you could go to one place online and fulfill your clinical, pre-clinical, or non-clinical trial needs with the press of a button?
Launching most clinical trials is hit-or-miss. Prices are all over the map. Quality is suspect. Staffing is a nightmare and patient populations are unpredictable. What if you could go to one place online and fulfill your clinical, pre-clinical, or non-clinical trial needs with the press of a button?
Four years ago, researchers at Cambridge Research & Development (www.cambridgerad.com) were in the process of starting their own clinical trials. Their experience with how the clinical trial community functioned led them to seek a better way. So they built ClinicalBid, a web-based portal that allows sponsors to quickly publish and manage multiple clinical trial proposals from the comfort of their office. No more chasing down vendors or the management nightmare of tracking proposals and bids. Sponsors can communicate with more than 200 (and growing) CROs in a single location.
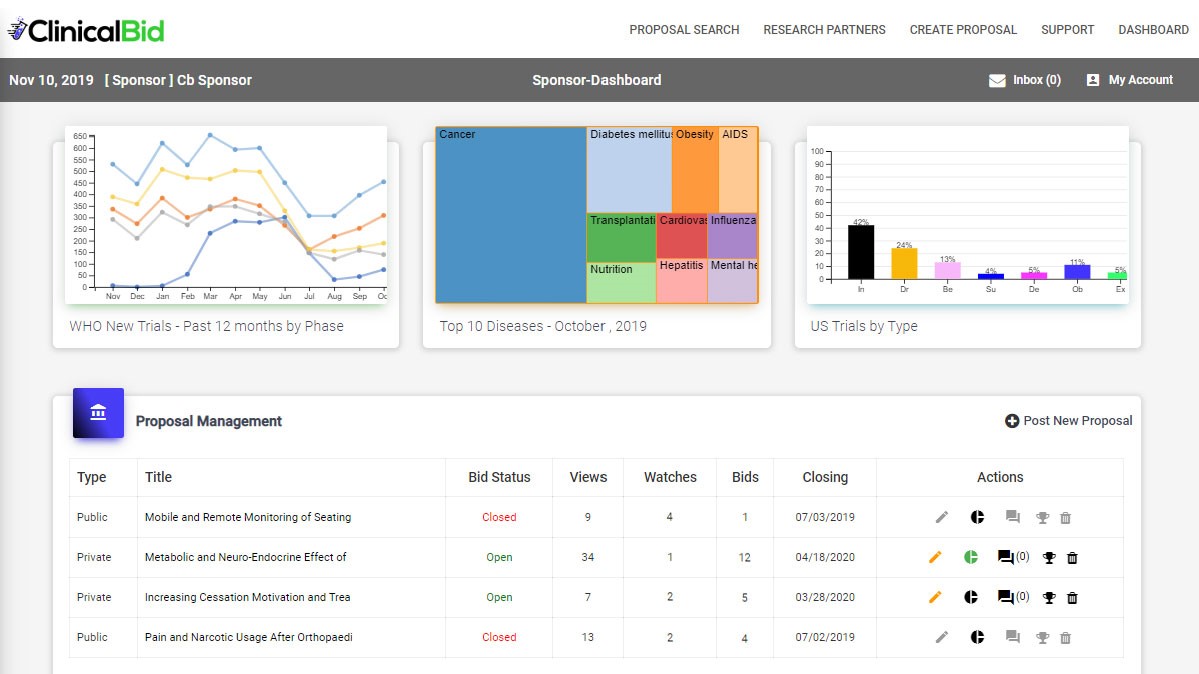
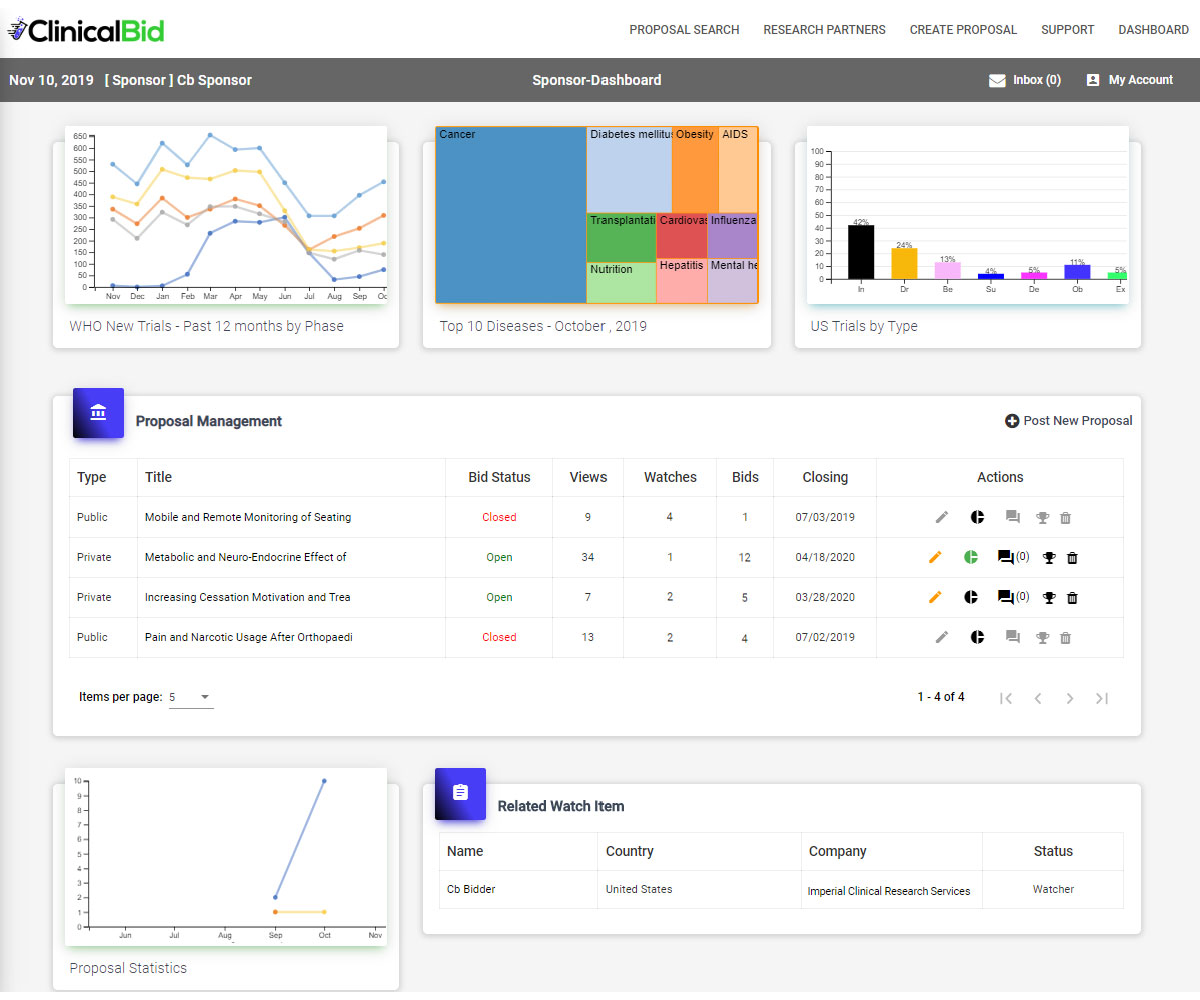
Sponsors can choose to post their trial proposals publicly or privately (by invitation only). Partners can then respond to posted proposals or set alerts (watches) for proposals they may be able to fulfill. In keeping with normal proposal lifecycles, there is an open Q&A period followed by Partners submitting their bids. The Sponsor reviews the bids and selects a winner.
In addition to driving down costs, ClinicalBid also helps Sponsors quickly locate trial participants using their “one-click” patient recruitment portal which taps into ClinicalBid’s patient population partnership network.
ClinicalBid is free for Sponsors and principal investigators with no cost to post a proposal. For CROs, payment is success-based. CROs and research institutes can sign up and submit bids for free, however, if they are selected as the winner, they pay a 3% success fee to ClinicalBid.