While other drug categories have matured, hypoglycemics remain one of the fastest growing categories in the sector. Diabetes prevalence is on the rise, and the industry reports a record 235 new medicines are currently in development for this high-need-to-treat condition.
With today’s competitive market and robust pipeline, manufacturers of existing and future hypoglycemics must understand the evolving payer market to maintain—or gain—optimal access for their brands.
MANAGED CARE COMPLEXITIES
Managed care customers are interdependent and share overlapping roles, which makes it difficult for marketers to identify key influencers. In addition are the regulatory, marketing, and disease prevalence differences among payer types. These also affect brand-access decisions to varying degrees. Successful marketers must embrace these complexities to understand the relationships between managed care customers.
The growing restrictions, formulary tiers, and myriad benefit designs create a complex formula for determining access, taking into account:
- Reimbursement, including copays, coinsurance, and benefit coverage.
- Restrictions, such as prior authorizations and step edits.
- Management, including patient compliance, guidelines, pay-for-performance, and disease management.
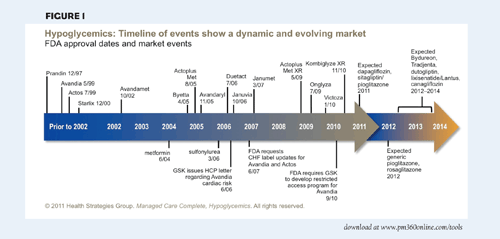
FIGURE 1
HOW PAYERS MANAGE HYPOGLYCEMICS
Growing disease prevalence and high utilization drive payers to prioritize hypoglycemics management. Thanks to the significant medical costs associated with untreated diabetes, however, the category remains largely unmanaged today. This offers brands broad formulary access with limited restrictions on utilization (e.g., step edits and prior authorizations).
In the near future, though, more payer policies may promote first-line use of lower-cost therapies, preparing for the generic availability of thiazolidinediones (TZDs, also known as glitazones), additional therapeutic options, and new therapeutic classes.
FIGURE 2
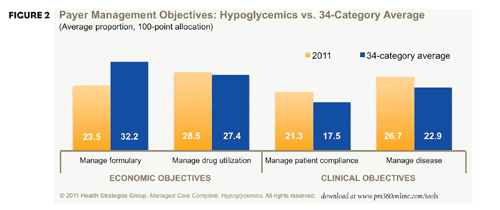
The shifting landscape within the market raises new questions for marketers of hypoglycemic brands including:
- What is the likelihood that plans will narrow options among preferred dipeptidyl peptidase-4 (DPP4) inhibitors or glucagon-like peptide-1 (GLP-1) analogs in exchange for additional rebates?
- What is the potential impact of new agents, including multiple DPP4s, GLP-1s, and sodium-glucose transport-2 (SGLT2) inhibitors? Are plans looking for leaders to emerge in each class?
- What innovative contract terms are needed to gain preferred access?
- How are managed care customers using prior authorizations and step edits to manage utilization?
- How critical is it for a product to demonstrate superiority during clinical trials?
- What type of support are manufacturers offering to help payers manage diabetes in their member populations?
- What product attributes are most influential in gaining preferred access?
OPTIMIZE BRAND ACCESS
Once manufacturers understand the complex factors that determine brand access in this important market, they can develop tailored, payer access strategies to maximize revenue for existing and potential new-to-market brands. They may consider a variety of access-optimization strategies, including:
- Manufacturers of inline brands facing pressure from payers to retain preferred access should avoid providing additional discounts, as payers are unlikely to demote brands.
- Although they primarily affect Avandia, cardiovascular safety concerns with TZDs are driving utilization to other classes. Manufacturers of DPP4s and GLP1s should emphasize safety advantages to payers to avoid potential step edits when generic TZDs enter the market in 2012.
- Absent strong clinical differentiation, manufacturers of pipeline DPP4s must generate early provider demand and price competitively.
Opportunity for preferred access may be available in smaller, more tightly controlled segments (i.e., regional independents, Blues affiliates) seeking to narrow preferred access in exchange for rebates. Most national plans, however, will likely limit preferred access to class-leading Januvia and new-to-market Onglyza. For this larger plan segment, manufacturers should leverage existing brands (portfolio contract) to improve access.
- To encourage drug therapy and limit future medical system costs, payers are accepting of new hypoglycemic classes. As a result, most plans will likely provide broad access to the first-in-class SGLT2 inhibitor. The first-to-market manufacturer should resist providing deep discounts and instead focus on generating provider demand.
CRITICAL MARKET EVENTS THAT MAY IMPACT PAYER ACCESS
The hypoglycemics market has been quite active in recent months. In January 2010, the FDA approved Novo Nordisk’s once-daily Victoza (liraglutide). In 2011, AstraZeneca and Bristol-Myers Squibb plan to file for FDA approval of dapagliflozin, a first-in-class SGLT2 inhibitor. And in 2012, Actos (pioglitazone) and Avandia (rosaglitazone) will likely lose exclusivity. (See Figure 1 timeline)
Between 2012 and 2014, additional agents that may gain FDA approval include:
- Two once-weekly injectable GLP-1s: Genentech’s taspoglutide and Amylin’s Bydureon (exenatide);
- Two more DPP-4 inhibitors: Boehringer Ingelheim’s Tradjenta (linagliptin) and Forest’s dutogliptin;
- Lixisenatide (sanofi-aventis), a once-daily GLP-1 combined with basal insulin (Lantus);
- Canagliflozin (Johnson & Johnson), a potential second-to-market SGLT2 inhibitor.
To download Market Drill-Down Tools for this issue’s topic, go to www.PM360online.com/tools
REACH OUT TO OUR MANAGED MARKETS ACCESS EXPERTS! Email your Managed Care question to Health Strategies Group for deep analysis. All queries will be considered for a future column. Email your question to Tom Martin, our Managed Care expert, for the answer.