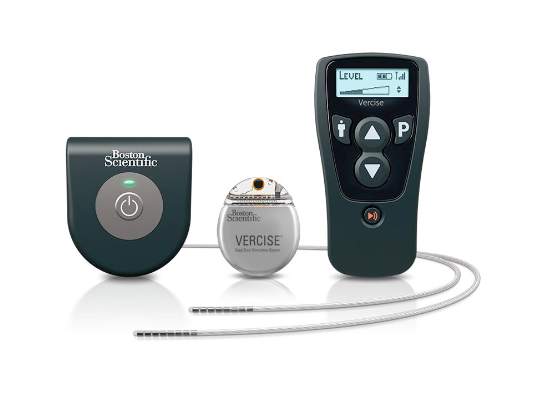
The Food and Drug Administration has approved the Vercise Deep Brain Stimulation (DBS) System to treat the symptoms of Parkinson’s disease.
The approval is based on as-yet unpublished results of the INTREPID trial, which successfully met its primary endpoint of mean change in waking hours with good symptom control, according to an announcement from the device’s manufacturer, Boston Scientific. The INTREPID study is the first multicenter, prospective, double-blind, randomized, sham-controlled study of DBS for Parkinson’s disease in the United States and enrolled in 292 patients at 23 sites.
A European multicenter, prospective, single-arm study in Europe, called VANTAGE, provided additional data for the approval ( Lancet Neurol. 2015;14[7]:693-701 ). That 40-patient study showed 63% improvement in motor function at 52 weeks from baseline as measured by the Unified Parkinson’s Disease Rating Scale III, as well as improvements in quality of life and medication usage.
The implantable pulse generator of the device is “the smallest, rechargeable DBS device available in the U.S.,” according to Boston Scientific, and can independently control the amount of current delivered by each of the electrodes on the implanted leads, allowing them “to work together to address common challenges in DBS therapy such as fluctuations in symptoms and the progressive nature of the condition by offering more adaptable delivery of stimulation.”
“The Vercise DBS System changes the landscape of what physicians can do to help improve the quality of life for people living with Parkinson’s disease,” Jerry Vitek, MD, PhD , professor and chair of the department of neurology at the University of Minnesota, Minneapolis, and coordinating principal investigator for the INTREPID study said in the Boston Scientific announcement. “This system provides an ability to sculpt the current field in the DBS target using novel technology that offers flexibility in programming. This flexibility allows us to target different regions of the subthalamic nucleus, which we believe will improve outcomes while reducing side effects.”