PM360’s Innovations Issue was established six years ago to serve as a comprehensive guide of the year’s most innovative Companies, Divisions, Startups, Products, Services, and Strategies. PM360 received more than 100 total submissions this year and picked the best in each category.
Here are our selections for the most innovative companies of 2017.
AdhereTech
Dan Gusz, Head of Product & Strategy
sales@adheretech.com
 AdhereTech delivers customized support programs to patients taking specialty medications. To deliver these programs, the company manufactures and supports smart connected pill bottles, which patients receive from the specialty pharmacy (free for patients, free for the pharmacy, and sponsored by pharma). AdhereTech has active partnerships with many of the world’s largest pharmacies, pharmaceutical companies, and healthcare firms. These programs allow patients across the globe to use AdhereTech’s patented smart pill bottles, each and every day.
AdhereTech delivers customized support programs to patients taking specialty medications. To deliver these programs, the company manufactures and supports smart connected pill bottles, which patients receive from the specialty pharmacy (free for patients, free for the pharmacy, and sponsored by pharma). AdhereTech has active partnerships with many of the world’s largest pharmacies, pharmaceutical companies, and healthcare firms. These programs allow patients across the globe to use AdhereTech’s patented smart pill bottles, each and every day.
AdhereTech’s bottles automatically collect and analyze adherence data in real-time. As patients use the bottles, adherence data is wirelessly sent from the bottles to AdhereTech’s servers. The system then automatically compares what patients are doing to what they should be doing. If a dose is missed, AdhereTech reminds the patient and/or caregiver via a series of customizable features, such as automated phone calls or text messages as well as on-bottle lights and chimes. But AdhereTech also goes beyond simple reminders. If the system notices certain patterns of non-adherence, it can solicit feedback from patients via text or phone call, asking them why a dose was missed.
AdhereTech’s average user is over 70 years old, and one-quarter of these patients don’t own a cell phone. That’s why AdhereTech’s bottles are built specifically with the non-adherent patient’s user-experience in mind. The wireless cellular technology allows their bottles to connect and work right out of the box, with zero setup. Additionally, the bottles are used just like normal pill bottles, down to the standard child-resistant cap.
AdhereTech collects de-identified, aggregated datasets that include information and analysis regarding: Patient dosage patterns, why patients stop taking the medication, when side-effects occur during a patient’s treatment (with AE reporting), predictive analytics, and much more.
Caribou Biosciences, Inc.
Gregory Kelley, Senior Vice President, Feinstein Kean Healthcare
gregory.kelley@fkhealth.com
 Caribou Biosciences is pioneering the development of CRISPR gene editing technology across human therapeutics, agriculture, industrial biotech, and biological research. Since its inception, the company’s singular focus has been to advance and refine this revolutionary technology, with the ultimate goal of developing new products that have the potential to deliver profound benefits to both human health and society as a whole.
Caribou Biosciences is pioneering the development of CRISPR gene editing technology across human therapeutics, agriculture, industrial biotech, and biological research. Since its inception, the company’s singular focus has been to advance and refine this revolutionary technology, with the ultimate goal of developing new products that have the potential to deliver profound benefits to both human health and society as a whole.
As a result of the company’s extensive IP portfolio, world-class research team, and key industry collaborations, Caribou has emerged as a leader across the CRISPR space. Since its founding in 2011, the company has raised more than $40 million in venture funding and has secured key strategic partnerships with industry leaders. In the area of human gene and cell therapies, Caribou co-founded Intellia Therapeutics in 2014 in order to utilize the power of CRISPR technology to develop curative medicines for a host of serious diseases.
Caribou has been focused on conducting cutting-edge research to overcome some of the most significant technical challenges facing CRISPR companies today. With key papers published in Nature Methods and Molecular Cell, the company is continuing to operate at the forefront of scientific discovery and has been responsible for making significant advances that will pave the way for commercialization of CRISPR products.
Looking to the future, the gene-editing technology market is projected to grow to $3.5B by 2019. As one of the pioneers of the field, Caribou is harnessing the power of CRISPR technology to accelerate the pace of biological research and to address devastating genetic diseases.
electroCore
Lauren Amodeo, Vice President of Marketing
lauren.amodeo@electrocorellc.
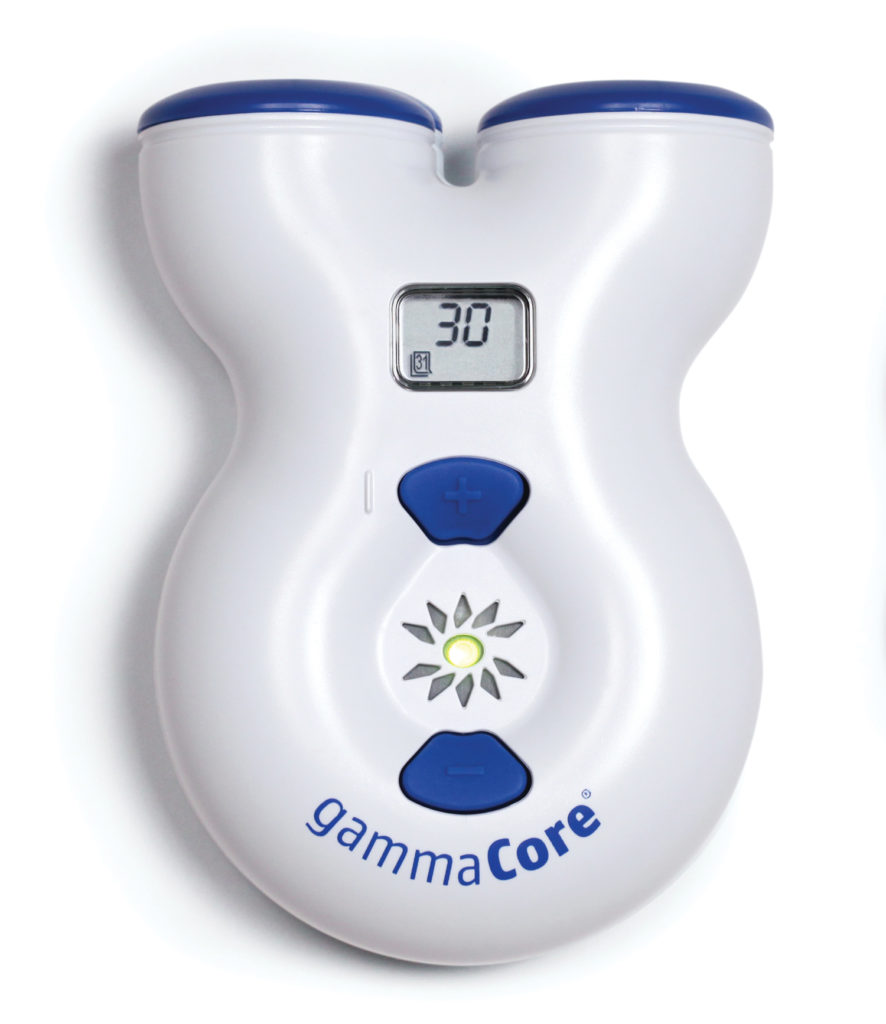 electroCore was founded in 2005 with the belief that neuromodulation could be advanced and used in novel ways in conditions for which there was an unmet patient need. Given that different modalities of neuromodulation—primarily implants—had previously proven to be effective and safe, the company recognized a need and a great opportunity to take neuromodulation to the next level and develop the first ever non-invasive vagus nerve stimulator (nVNS).
electroCore was founded in 2005 with the belief that neuromodulation could be advanced and used in novel ways in conditions for which there was an unmet patient need. Given that different modalities of neuromodulation—primarily implants—had previously proven to be effective and safe, the company recognized a need and a great opportunity to take neuromodulation to the next level and develop the first ever non-invasive vagus nerve stimulator (nVNS).
In April 2017, the FDA released gammaCore®, the first hand-held, nVNS therapy to treat the acute pain associated with episodic cluster headache in adult patients. Cluster headache is a rare type of excruciating headache that affects about one to two people in every 1,000. Its pain can be even more debilitating than a migraine, and it predominantly affects men, with symptoms typically manifesting by the age of 30. This was an important advancement in the cluster headache space, as it does not have the side effect profile or dose limitations of commonly prescribed treatments.
In July 2017, the device became commercially available in the U.S. through the gammaCore Patient Registry, a non-research registry intended to examine the patient experience with gammaCore. This marked the first U.S. clearance and marketed product for electroCore. In September 2017, electroCore revealed initial pivotal trial data for gammaCore with the intentions of expanding the device’s current indication. The findings were clinically significant, supporting a potential FDA submission for an indication in migraine in the near future. This could be a key development in the migraine space, as migraine disrupts the lives of more than 30 million Americans with an incidence of approximately 18% in women, 6% in men, and 8% in children.
Klick Health
Alec Melkonian, SVP, Client Engagement
connect@klick.com
 Klick Health, the world’s largest independent health marketing and commercialization agency, is always in Beta, always exploring and experimenting, and always finding new and exciting ways to use its capabilities to advance the health sciences industry and help HCPs and patients.
Klick Health, the world’s largest independent health marketing and commercialization agency, is always in Beta, always exploring and experimenting, and always finding new and exciting ways to use its capabilities to advance the health sciences industry and help HCPs and patients.
The company’s digital innovation lab, Klick Labs, helps life science organizations advance healthcare through the application of emerging concepts and technologies. Klick Labs offers a full range of innovation services, including primary research, ideation, design, prototyping, and in-market validation under a Lab as a Service (LaaS) model.
Over the past year, Klick has focused on two major industry innovations. First, after launching Circulation in September 2016, the company focused on rolling-out this first-ever customizable, patient-centric digital transportation platform that integrates within both healthcare systems and Uber’s API. Working with Dr. John Brownstein and the other co-founders of Circulation, Klick set its sights on improving the state of Non-Emergency Medical Transportation (NEMT) and quickly implemented Circulation at 1,000 top-tier hospitals, clinics, community health centers, and other healthcare facilities nationwide. In July, the company also secured $10.5 million in Series A financing to further expand Circulation across the country.
Second, in April 2017, Klick introduced the emerging field of tele-empathy with the unveiling of the SymPulseTM Tele-Empathy Device, the first device, which wirelessly records and transmits Parkinson’s tremors to help increase clinical empathy. Increased clinical empathy is associated with better patient outcomes, and, according to Dr. Helen Riess, Associate Professor of Psychiatry at Harvard Medical School and Founder of Empathetics, Inc., “Improving clinical empathy should be at the forefront of institutional missions and interventions are needed to achieve these critical healthcare goals.”
Lockwood
Christine Hinkle, Chief People Strategist
christine@thelockwoodgrp.com

 Lockwood has been steadily growing 40% year over year in revenue and staff for the last 10 years due to an innovative approach to talent recruiting. The company credits its success to four key tactics:
Lockwood has been steadily growing 40% year over year in revenue and staff for the last 10 years due to an innovative approach to talent recruiting. The company credits its success to four key tactics:
- Front loading talent: The company hires employees before they’ve won client business to allow them the time to get familiar with Lockwood. Adding people creates bandwidth and that bandwidth is what allows Lockwood to do a number of things for clients such as take on extra work from clients and meet aggressive deadlines.
- Don’t hire by position: When hiring by position, an organization and its hiring managers have tunnel vision about whether or not a candidate is a fit for a role with specific capabilities and responsibilities. Instead, Lockwood uses guiding principles for all client-facing roles. For instance, they are only interested in talent that has at least three years of experience working in a medical communications agency.
- Not bound by geography: 60% of the company’s workforce is 100% remote-based across 19 different states. This model allows teams to have a balance of work and life by eliminating the need for a long commute to the office.
- Fulfilling promises: Lockwood makes a lot of promises to candidates after they are brought on board, such as opportunity for career growth, a culture that values them and their ideas, and staffing plans that have bandwidth so that they aren’t working week over week 60 to 70 hours without reprieve. The company is dedicated to making these promises more than just lip service—and exceed new hires’ expectations.
MyHealthTeams
Joe Marziani, SVP of Partnerships
joe@myhealthteams.com
 The lines of communication have never before been so open between pharma companies and their consumers, yet many companies still struggle to understand the patient perspective. The 28 social networks created by MyHealthTeams are fast becoming a powerful tool for closing this gap.
The lines of communication have never before been so open between pharma companies and their consumers, yet many companies still struggle to understand the patient perspective. The 28 social networks created by MyHealthTeams are fast becoming a powerful tool for closing this gap.
MyHealthTeams creates social networks for people living with a chronic condition—ranging from common conditions such as migraine and Alzheimer’s to rare conditions such as pulmonary hypertension (and many others, including Crohn’s, colitis, rheumatoid arthritis, and lupus).
In sum, the company’s 28 social networks address 90% of the chronic condition population. More than 1.2 million members rely on their MyHealthTeams’ community for information, resources, lifehacks, and support. Engagement rates are approximately 10 times better than the average health-related website—with 30% to 60% of MyHealthTeams’ members returning monthly.
More than two dozen pharma and biotech brands have teamed with MyHealthTeams to create valuable patient education resource centers and to gain real-world insights into the patient experience. MyHealthTeams works with partners at every stage of content strategy, research, development, and commercialization of new therapies.
More than 95% of MyHealthTeams members are diagnosed patients, with caregivers making up the remaining 5%. They share extensive information about their chronic condition experience, such as when they were diagnosed, what symptoms they experience, which drugs they take, what their baseline knowledge of treatment options is, and what side effects they’ve experienced.
This enables pharma marketers to educate a highly engaged audience efficiently and provides confidence that the message is reaching the right people in context, while they are searching for answers about their condition. MyHealthTeams develops customized approaches—branded and unbranded—to close the knowledge gap among patients regarding their treatment options.
Praxis Communications
Tricia Barrett, SVP, Managing Director
tbarrett@gopraxis.com
 Praxis Communications is a full-service patient recruitment and retention organization that specializes in optimizing study enrollment for research studies with award-winning creative and superior execution.
Praxis Communications is a full-service patient recruitment and retention organization that specializes in optimizing study enrollment for research studies with award-winning creative and superior execution.
Praxis’ innovation lies in its approach to patient recruitment and retention—using scientific data to inform creative art. The Praxis analytics team is trained in social listening, linguistic analysis, predictive analysis, in-depth interviews, and more. These disciplines are applied early on in the recruitment process to identify the words, images, concepts, and approaches that our target patient uses to relate to her situation and condition.
From there, the Praxis creative team reviews all of the analytical data and insights gleaned, and designs a campaign—posters, ads, logos, brochures, cards, etc.—that speaks directly to the patient in her own terms. Within the past year alone, Praxis has used analytics to inform creative work for more than 75 potential and current clients.
Having invested in Sysomos for years, in late 2016 Praxis began also investing in Synthesio, an in-depth global social listening platform, as well as in an upgraded Statistical Package for the Social Sciences (SPSS), which includes predictive modeling and advanced statistical analysis. These upgrades mean that the data is cutting-edge, and the results are increasingly tailored.
Even with a proper analysis in place, it requires a special creative professional to be able to appropriately capture the essence of raw human emotion in a persuasive piece of creative work. That is why the greatest challenge Praxis has faced in developing its science-informed art innovation has been in painstakingly curating the right team of focused, experienced, and dedicated professionals. But, with a strong team in place—and growing fast—this innovation has taken hold and created a bright future for patient-centered clinical research recruitment.
QoC Health
Raymond Shih, President
solutions@qochealth.com
 In 2017, the mobile health market is expected to reach $24.2 billion worldwide. The digital health market is expected to reach $233.3 billion by 2020, driven particularly by mobile health. In the future, it is not hard to envision all smartphone users using healthcare applications eventually. This is where QoC Health comes in. The company helps innovators take their digital health ideas to scale and/or commercialization with its patient engagement technology platform.
In 2017, the mobile health market is expected to reach $24.2 billion worldwide. The digital health market is expected to reach $233.3 billion by 2020, driven particularly by mobile health. In the future, it is not hard to envision all smartphone users using healthcare applications eventually. This is where QoC Health comes in. The company helps innovators take their digital health ideas to scale and/or commercialization with its patient engagement technology platform.
The platform’s capabilities include: Patient monitoring, analytics, educational materials, secure communications, decision aids, planning tools, and turnkey integration with existing medical records. Tools built using QoC Health’s platform have peer-reviewed, published results demonstrating both improvements to patient outcomes and experience and cost savings using its technology.
What makes the product truly unique is that it’s not an app, but a true development platform that enables rapid prototyping of mature digital health solutions that can immediately scale in multiple geographies (currently U.S., E.U., and Canada). Furthermore, the platform enables enterprise features such as turn-key EMR integration, multi-language management, and direct licensing.
The tools created through QoC Health help the healthcare industry in three ways:
- Improves patient outcomes: By building tools to provide clinicians with new, accurate data, they can make better, proactive interventions.
- Improves patient experience: Tools created through the platform regularly have patient satisfaction ratings over 95%.
- Reduces the cost of care: For example, a post-surgical tool developed by the company was shown to reduce costs by 65% in its first year.
Bayshore, one of Canada’s largest home care providers and now one of QoC Health’s largest investors, offered this testimonial: “We get the opportunity to evaluate many digital health companies. QoC Health offers a combination of perfect-fit solutions with off-the-shelf simplicity that is unique in the market.”
Squint Metrics LLC
Marc Porter, Managing Director
marc.porter@squintmetrics.com
 Squint Metrics LLC was founded to address the significant deficiency in real-world outcomes data as it relates to chronic, disabling disease states/conditions. The company places an emphasis on balancing the collection of as many relevant data points as possible—without becoming a burden to patients, caregivers, and healthcare providers—through electronic patient-reported outcomes (ePRO) programs. The platform allows rapid analysis and response to changing disease stability outcome metrics as well as treatment utilization trends by moving beyond raw quantification to a deeper understanding of clinical management.
Squint Metrics LLC was founded to address the significant deficiency in real-world outcomes data as it relates to chronic, disabling disease states/conditions. The company places an emphasis on balancing the collection of as many relevant data points as possible—without becoming a burden to patients, caregivers, and healthcare providers—through electronic patient-reported outcomes (ePRO) programs. The platform allows rapid analysis and response to changing disease stability outcome metrics as well as treatment utilization trends by moving beyond raw quantification to a deeper understanding of clinical management.
In order for population health data to become truly translatable to individual care, patients, care partners, and healthcare professionals must work together as one cohesive team. Squint ePRO programs leverage user-friendly and validated surveys, phone interactions, and available wearable technology to capture essential outcomes and relevant lifestyle endpoints. The user-personalized dashboards enable local, regional, and national population comparison across demographic, disease status, and treatment parameters. The result is on-demand data which supports improved disease and treatment knowledge, enhanced HCP-patient communication, and better long-term patient outcomes.
To date, the company has launched two ePRO products: Gut Check for IBD which launched in partnership with Janssen Biotech in 2015 and MS Care Connect for multiple sclerosis which launched in December 2016 in partnership with leading neurologists. Currently, both programs have greater than 85% user persistency, EHR integration capability, and enjoy rapidly growing user populations. With fully customizable partner dashboards, Gut Check and MS Care Connect provide commercial partners invaluable strategic direction, publishable outcomes data, and an efficient and effective electronic data capture (EDC) platform for potential application in clinical trials. Squint Metrics is currently developing programs in several additional categories planned for 2018 launch as they seek to further support improved patient care.
Startupbootcamp Digital Health Miami
Natasha Linz, Marketing Manager
natasha.linz@startupbootcamp.org
 Startupbootcamp (SBC) Digital Health Miami supports and scales extraordinary companies transforming healthcare and eradicating disparities. To date, the company has invested in 12 companies from across the U.S., Latin America, and Israel. Some of the startups within SBC Digital Health Miami’s portfolio include: Aces Health (the first end-to-end clinical trial management app for patients and researchers); Babyscripts (the first mobile, clinical solution to seamlessly provide remote monitoring of an OB’s patient population in-between visits); and CareAngel (an artificially intelligent nurse assistant).
Startupbootcamp (SBC) Digital Health Miami supports and scales extraordinary companies transforming healthcare and eradicating disparities. To date, the company has invested in 12 companies from across the U.S., Latin America, and Israel. Some of the startups within SBC Digital Health Miami’s portfolio include: Aces Health (the first end-to-end clinical trial management app for patients and researchers); Babyscripts (the first mobile, clinical solution to seamlessly provide remote monitoring of an OB’s patient population in-between visits); and CareAngel (an artificially intelligent nurse assistant).
In partnership with the Knight Foundation, Miami Children’s Hospital, Accenture, SBC provides funding, customer contract opportunities, a mentor network of industry experts, executives and top tier investors, workspace, and a comprehensive suite of services so that entrepreneurs can focus on doing what they do best: Build game-changing businesses.
This past month 13 companies were chosen from more than 300 applicants to join SBC’s second cohort: B.well, BrainFX, Cybexys, Empower Capital, Epharmix, FRND, HealthTensor, NarrativeDX, NeuraMetrix, Quick’rCare, SaveMyScope, Twiage, and Wellth.
The program kicks off on January 15, 2018 when the startups will relocate to Miami. The program offers participating companies partnerships with leading hospital systems, insurers, and pharma to accelerate and scale their businesses, extensive mentoring from more than 150 healthcare entrepreneurs, investors, and executives in addition to six months of free office space and seed funding.
Key focus areas for SBC include: Artificial intelligence, population health management, behavioral health, telemedicine, payer administration and analytics, virtual and augmented reality, big data and analytics, remote patient monitoring, EHR and clinical workflow, and personalized and preventative medicine.
Founded in 2010, SBC is an award-winning global network of industry-focused innovation programs with more than 20 programs in Europe, Asia, North & South America, MENA, and Africa.









