PM360’s Innovations Issue, established five years ago, serves as a comprehensive guide to our readers, providing a glimpse at the year’s most cutting-edge: Companies, Divisions, Startups, Products, Services, and Strategies.
Here are our picks for the most innovative products of 2016, which include software, apps, programs, and anything else designed to improve how those working in the industry do their jobs.
Avatar
Xcell Biosciences
Martin Pieprzyk, Executive Vice President of Sales and Marketing
From drug discovery to disease modeling and pathway interrogation, scientists analyzing cells need the most accurate data about gene and protein expression. Unfortunately, too many cell-based assays are performed on cells that no longer express the same genes and proteins as cells in their natural environment. More and more evidence implicates traditional cell culture systems, which fail to accurately mimic cells’ native environment and lead researchers to produce misleading data. The Avatar system is a next-generation incubator that better models the cellular microenvironment and has been shown in many studies to culture and grow cells that more accurately reflect true biology.
Avatar, introduced in March 2016, is a revolutionary system for cell-based assays that replicates the in vivo microenvironment of cells to deliver reproducible, biologically relevant results. It was designed to create an environment that could be more carefully tailored to any type of cell, and thus increase success rates for maintaining and expanding difficult-to-grow cells.
This innovative culturing system improves on current incubators by incorporating important biological traits—hypoxia and pressure—as fully customizable settings. By using adjustable settings for oxygen and pressure levels in addition to the usual temperature and carbon dioxide levels, the Avatar system allows scientists to recreate the native microenvironment of their samples and to generate more physiologically relevant results. The system includes a bench-top and stackable instrument as well as consumables for a number of applications such as cancer research, biomarker discovery, lead candidate selection and optimization, stem cell research and regenerative medicine, and immunotherapy drug development.
 Axium Neurostimulator System
Axium Neurostimulator System
St. Jude Medical
Justin Paquette, Senior Manager of Public Relations
St. Jude Medical has a 40-year legacy of developing innovative, cost-effective medical technologies that save and improve the lives of patients around the world. This legacy is exemplified by St. Jude Medical’s new dorsal root ganglion (DRG) neurostimulation technology, affording patients suffering with chronic pain superior relief compared to conventional neurostimulation.
The Axium Neurostimulator System (http://bit.ly/2fCEFt7) is the only FDA-approved system specifically designed to stimulate the DRG, which has been clinically proven to provide superior pain relief over conventional tonic spinal cord stimulation (SCS) in patients with neuropathic chronic intractable pain associated with complex regional pain syndrome (CRPS), a chronic pain condition most often affecting one of the limbs. DRG stimulation expands the range of neurostimulation options physicians can offer, including the ability to treat the specific areas of the lower limbs where pain occurs, and may help physicians significantly improve the lives of patients with CRPS, which has typically been very challenging to treat.
The Axium Neurostimulator System, approved by the FDA in February 2016, is the result of nearly a decade of R&D. The effectiveness of DRG stimulation was proven in the ACCURATE study, the largest randomized neurostimulation clinical trial comparing conventional tonic SCS and DRG stimulation to treat CRPS. The study was conducted by physicians at more than 20 U.S. centers as well as by physicians across Europe (where it has been available since 2011). The findings demonstrated that DRG stimulation was clinically superior to conventional tonic SCS in treating lower extremity pain (foot, knee, hip, groin) associated with focal chronic intractable pain due to CRPS.
AzaadHealth
AzaadLabs
Syed Abrar, CEO
A recent survey by GFK (http://bit.ly/2fBbkSF) revealed that 33% of the worldwide population track and monitor their health online. This demonstrates the attraction health monitoring has for a wide audience. But for the longest time, most of the population has been unable to access their health information from healthcare providers such as hospitals, pharmacies, and laboratories.
AzaadLabs (www.azaadlabs.com) believes it’s high time to solve this problem. Its product, AzaadHealth (www.azaadhealth.com), fetches and aggregates an individual’s health information from hospitals, pharmacies, laboratories, and wearable fitness devices. This will let people view and monitor their own and their loved one’s complete health in one place. Users can also securely share their health profile with other hospitals and doctors.
In this age of big data, getting health information to hospitals and patients is not enough. The information must also help people to better understand their health. AzaadHealth’s analytics engine takes care of this by employing machine learning and predictive analysis, which provide in-depth health insights to hospitals and individuals.
Conceived earlier this year at Draper University, AzaadHealth was born out of the sheer pain of losing their co-founder’s brother to liver cancer. AzaadLabs was initially a software development firm which provided app development services to off-shore clients, which allowed the company to fund and bootstrap AzaadHealth.
Now, AzaadHealth is incubated at Nest i/o (http://thenestio.com), a tech incubator in Pakistan managed by P@SHA (http://pasha.org.pk) and funded by Google, Samsung, and the U.S. consulate. The company also became a registered vendor partner for the Australian Digital Health Agency, which allows it to collaborate with the Australian government in promoting mobile health. The plan is to release AzaadHealth for Australia and U.S. by the end of December 2016.
Backstage® Duet
closerlook, inc.
Michael Tapson, Chief Technology Officer
Humans create 2.5 quintillion bytes of data every day—and 90% of all data ever produced has been made in the last two years. So it’s no surprise that the pharmaceutical industry is experiencing an unprecedented increase in data collection. But what do we do with all of that data? Collecting it is one thing. Aggregating and analyzing the data is another.
Enter Backstage® Duet, a cloud-based insight discovery application that helps marketers gain a deeper understanding of their audience and marketing performance. Backstage® Duet, which closerlook will roll out in early 2017, gives clients a 360° view of HCP targets and the ability to apply metrics to optimize campaign management.
It’s built by marketers for marketers. Its easy-to-use interface provides pharma-specific multichannel insight to enable smart campaign optimization, and it provides customer and campaign-level insight across all channels. By purposefully limiting Backstage® Duet’s feature set to include audience generation, analysis, and reporting, closerlook was able to create a product that provides a clear value for its clients.
Backstage® Duet:
- Makes it easy to find the right audience.
- Provides a view into an HCP’s patient population.
- Reveals the messaging that has the most impact.
- Predicts outcomes before a marketing campaign is launched.
- Simplifies complex calculations into a single score to allow clients to make better marketing decisions.
- Provides easy-to-read summaries of a client’s most important data.
Ultimately, Backstage® Duet provides an easy-to-use multichannel hub for aggregating and analyzing data on all of a brand’s marketing initiatives, as well as proprietary KPIs.
CancerLandscape
Kantar Health
Cory Lewis, PhD, Manager, Oncology Commercial Development
The oncology market is rapidly changing. Thousands of compounds are in development and clinical trials that involve new mechanisms of action and new indications. Getting a handle on the sheer volume of data and reconciling it into a useable format to inform decision-making is a daunting and time-consuming process. CancerLandscape was developed to streamline Kantar Health’s internal data search and analysis processes, but the company soon found it also addressed its clients’ needs as they faced the same challenges.
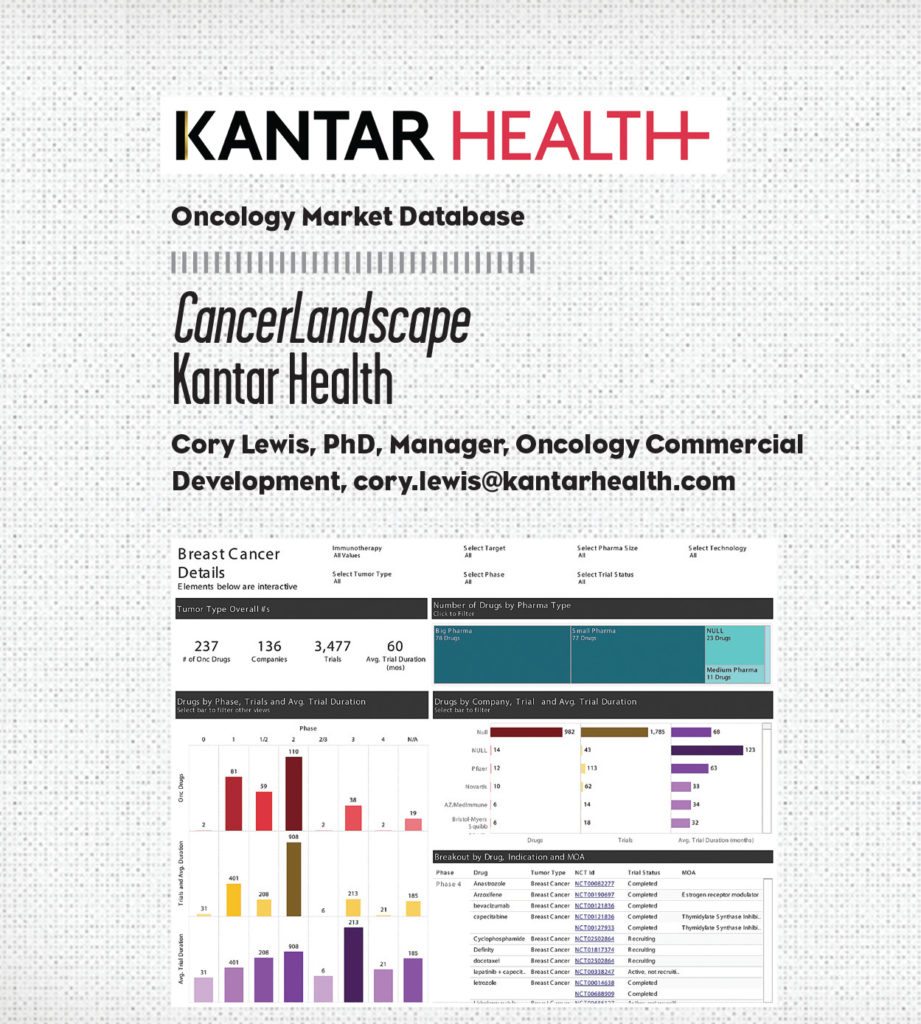
CancerLandscape aggregates many large oncology datasets, allowing for quick navigation of the data to better understand oncology drug opportunities and the competitive landscape. This web-based solution enables users to review more than 2,000 cancer drugs covering more than 175 tumor types. Each medicine is tracked by company, mechanism of action, tumor type, geography, and stage of clinical development.
While other data sources can be disparate and hard to navigate, the CancerLandscape tool takes the manual work away from clients. This results in increased efficiency—cutting through the clutter and delivering results in minutes instead of weeks. In addition, the platform can be operated across teams because it does not rely on a system-trained person.
The data visualizations within the platform allow users to quickly understand large, complicated datasets. Each output is unique and tailored through the clickable and exportable filters, while maintaining visual consistency across dashboards.
CancerLandscape assembles and analyzes oncolytics using both scientific and commercial filters. Kantar Health’s scientific team provides expert quality control and a systematic approach to ensure confidence in the data. CancerLandscape can also review, filter, and isolate preliminary agents to identify market attractiveness and target unique opportunities.
DrivenBI SRK
DrivenBI
Kathleen Douglas, EVP Global Sales
DrivenBI SRK redefines self-service Business Intelligence (BI) by removing the IT complexity associated with data analytics. Usable by business professionals in just a few days, DrivenBI delivers a cloud-native, self-service solution that does not require constant involvement from technical professionals and empowers users to be able to bring the data forward by themselves within the decision-making window and disseminate insights across all lines of business in a timely manner.
The SRK platform has been used by medical, pharma, and biosciences professionals responsible for marketing, operations, and sales. It can also be integrated into finance, inventory control, resource management, and corporate-wide KPIs.
DrivenBI SRK is more than just preparing, storing, and analyzing data, or building and sharing reports and dashboards. It also offers a set of features making the analysis results actionable in a centralized and well-organized manner. Tasks can be easily created no matter where the user is browsing in the system, with notifications automatically pushed to relevant parties through emails or text messages to remind them in real time about actions that need to be taken. And with DrivenBI’s new onboarding methodology, new users can see how analytics can help with their specific business needs in just a few days.
Furthermore, the Collaboration Hub in SRK allows users to automate processes and create immediate impact. They can set thresholds that, once exceeded, will automatically send out alerts to notify personnel when, for example, sales quotas are not met or profit level is at risk. And the automatically triggered alert contains a shortcut to quickly create a Task in SRK, so that the potential issues exposed by the analysis can be acted upon immediately.
mapt
watzan
Charles Benaiah, Chief Executive Officer
mapt began life as an app called ZEN. The premise was simple: People have more choices today than ever, but the solutions that power discovery haven’t kept pace. When it launched two years ago, ZEN was more about idea than execution.
Since then, watzan has learned plenty to help improve the offering. For instance, the average HCP who attaches their Twitter account to ZEN follows more than 320 people. Social matters. The company also learned what people want, who they want to follow, and myriad other things influencing how they choose content.
The company reimagined the product as a way to move people around content quickly and easily—like a map for content. Hence the new name: “mapt.” It has all the features of a social network—following, content discovery, and the uncanny ability to uncover like-minded people. In short, mapt puts people on a content discover journey powered by their interests and their unique social network.
However, while marketers want to be part of the customer journey and tell their story along the way, it’s not easy. This is especially true in pharma as the industry has certain rules about “wild west” media, such as Twitter and Facebook.
That is why mapt is “closed social.” You can show up and act on the content that watzan makes available, but you can’t bring content into the system. And yet, the platform still uses the information it learns from a person’s social media to personalize how they see content on the app. For instance, if it is being used at a conference, the sessions or speakers a user would most likely want to hear appear larger than the rest.
MedSked Mobile Smart Label
Med-Con Technologies, LLC
Anthony Londino, Vice President Marketing
MedSked (www.medsked.com) is a unique patient adherence solution used to support global clinical trials and other segments of the pharmaceutical industry. The core MedSked product is a self-adhering sleeve that is placed on the prescription bottle. Each sleeve has daily reminder tabs designed to help patients—at a glance—always remember to take the correct dose and track their medication use. And, because the MedSked sleeve can slide up and down the bottle, patients can still read the original prescription label at any time.
However, within the last year, Med-Con Technologies digitized the MedSked process, which makes it even more valuable for clinical trial use. The MedSked Mobile app enables patients to use their smartphones to scan the MedSked label sleeve, providing clinical teams with remote monitoring and time-stamp notification data, informing them whether every prescribed dosage has been taken or missed by the patient. The MedSked Mobile app can also send reminders to help patients take their medication on time and alarms when medications are missed.
Recently, the company took MedSked Mobile to the next level as now patient adherence data and management tools are available simply by applying its new peel-and-stick Smart Label to any bottle or other form of medication packaging.
In a recent independent study, a client found that improved adherence by patients using MedSked resulted in a 16% increase in Proportion of Days Covered (PDC). In another study of a multi-year clinical trial, the client projected savings of $6.40 per patient using MedSked with bottles instead of blister packaging—a total savings of $972,000 for the life of the trial.
My Story Storytelling Platform
Remedy Health Media
Jim Curtis, President, Advertising, Strategy and Operations
Remedy Health Media created the Live Bold, Live Now empowerment initiative to showcase curated inspirational stories of patients who have accomplished great things while overcoming the limitations of their condition. And research showed that visitors of a Live Bold, Live Now story took health-related actions due to the story. While hearing somebody else’s story is encouraging, the Remedy team recognized a need to be filled—all patients should have an opportunity to tell their own story. This led to the creation of My Story Storytelling Platform, a storytelling authoring tool that gives anybody with a health condition an outlet to seamlessly share their story.
My Story Storytelling Platform was launched (www.seemystory.io) using Remedy’s proprietary technology. This easy-to-use authoring tool allows people to expertly produce and publish stories using video, text, images, and audio to inspire and connect with others, which in turn provides a feeling of empowerment to the storyteller.
The tool’s interface allows users to view sample stories and prompts them through simple, easy-to-follow steps helping them develop their best emotional, engaging, and inspirational story. The platform leads people to share their own personal stories, but it also has many options and flexibility allowing increased story creativity and inspiration. The best of the best stories are selected and vetted by Remedy editors and then featured within Live Bold, Live Now: My Story Condition Centers.
So far, the results of the tool are proving effective to the viewers of these stories: 91% say they related to the story and 94% say they would recommend the story to others. Meanwhile, 100% of the storytellers report feeling empowered following sharing their story.
PatientPoint 360°
PatientPoint
Julie Cain, Vice President Program Management
Mobile apps for physicians have been available for some time now, but they tend to be “look up” tools, designed to allow the physician to search for symptoms, medications, or industry news. PatientPoint 360°, which launched in October 2016, is different—its goal is to partner with physicians to enhance the patient-physician interaction by pulling together the research news, anatomical tools, and patient education resources physicians need in one convenient app. Better yet, this information can be accessed immediately on their smartphones and tablets during their workday while in the exam room with the patient to facilitate a meaningful and effective conversation about a disease or condition to yield better outcomes.
With PatientPoint 360°, physicians can:
- View 3D anatomical models with patients in any exam room and have the option to email an image of the model directly to patients.
- Access the full library of PatientPoint educational brochures and email a link to patients.
- Stay up-to-date on industry news by reading selected curated articles from top medical journals both in the office and at home.
- “Favorite” articles, brochures, and models specific to their specialty for quick access.
- Search for condition-specific information via voice or text.
PatientPoint 360° will also help give time back to physicians and their staff. Every time they have to leave the exam room and search for information to provide to a patient adds to an already busy day. With PatientPoint 360°, this information is now available with a few taps of the finger.
Thumbprint
Lash Group
Anthony Martin, Senior Vice President – Client Strategy
In July 2016, Lash Group, a part of AmerisourceBergen, created Thumbprint, a set of individualized adherence solutions for manufacturers designed with the patient in mind. The personalized services offered through Thumbprint address predictive therapeutic and condition-related health risks and empower patients to be active participants in their own care. Thumbprint includes three unique service frameworks:
MyCommitment: This solution involves patient-directed communication, education, and outreach. MyCommitment promotes patient engagement and adherence by identifying barriers to care and meeting the individual needs of the patient. Features of this service model—which include a URAC-accredited telehealth nursing call center, patient-specific needs assessments, and tailored care plans—work together to help patients become empowered and engaged to increase commitment to therapy.
MyTransitions: Modifications in therapy, transition from hospital to home, and other life changes can make it difficult to maintain a patient’s commitment to therapy. The MyTransitions model works to ensure continuity of treatment despite transitions in care by thoughtfully and diligently connecting patients with their full healthcare teams, including caregivers and providers. The company’s approach to personalized support keeps patients and caregivers active and involved in their care by providing strategies and resources to overcome barriers and challenges that often lead to readmission or early therapy discontinuation.
MyConnection: MyConnection supports patients through focused disease state and product education interactions. To help avoid patient drop-off, targeted outreach campaigns anticipate patient needs related to disease progression or the onset of product side effects. For example, if a patient is prescribed a particular medication with delayed either positive or negative side effects—both of which may tempt a patient to discontinue use—MyConnection triggers outreach timed to coincide with the presentation of those side effects.
Veeva CRM Engage Product Family
Veeva
Jamie Morris, Vice President, Commercial Strategy
Life sciences companies have faced significant barriers to adopting digital capabilities and orchestrating interactions. Hindered by the limitations of siloed technologies, companies have struggled to manage digital engagements in CRM, gain actionable insights, and share compliant multichannel content.
Until now. The new Veeva CRM Engage family of applications provides a unified solution that makes digital engagement simple and compliant—unlocking important channels to life sciences companies for the first time. The Veeva CRM Engage applications include Veeva CRM Engage Meeting (making compliant online meetings easier and more convenient), Veeva CRM Engage Webinar (simplifying virtual events), and Veeva CRM Engage for Portals (providing HCPs self-directed online access from any device).
Embedded in multichannel Veeva CRM as part of a rep’s workflow, the Engage family allows easy reuse of approved content, minimizing compliance risk. All customer interaction data is aggregated and combined with insights gained across all channels for a complete customer view. This holistic understanding of physician needs will enable the industry to deliver more valuable interactions to HCPs, at the right time, through the right channels.
Veeva CRM Engage makes digital engagement viable for life sciences companies. As a result, HCPs can now get faster access to the information they need through the online and mobile channels they prefer—ultimately enabling deeper relationships between pharma and healthcare.
With Veeva CRM Engage, companies can:
- Improve customer engagement: Deliver the information customers need with compliant online content sharing and collaboration.
- Increase productivity: Ensure greater coverage, frequency, and reach with the same resources.
- Ensure consistency: Enable a coordinated experience across brands and channels.
- Strengthen compliance: Built-in controls for managing and displaying approved content minimize risk.