The Quantified Self (QS) movement, or lifelogging as some people call it, just keeps picking up steam as more wearable devices and mobile health (mHealth) solutions continue to flood the market—allowing people to keep track of how many steps they take, how well the sleep, how many calories they consume, how their workouts are progressing and much more. Some of the more popular wearables currently available include the Nike+ FuelBand, Fitbit and Jawbone UP. People use these devices and others to collect data to help improve their daily lives, find the best ways for them to stay fit, manage their chronic diseases, etc. However, despite the fact that Wired Magazine editors Gary Wolf and Kevin Kelly coined the term “quantified self” in 2007, this concept has been around for a while.
“Recall going on a body scale?” asks Juliana Chua, Principal of Zensorium Business Group, a company dedicated to empowering individuals to elevate their quality of life through a breed of innovative sensing devices such as the Tinké. “The movement of Quantified Self is not new,” Chua explains. “However the difference is the technology, which now enables us to quantify metrics we were previously unable to. With this increasingly popular global movement, it helps us all make better decision, set goals to gain control, self-explain and review with a critical eye to monitor progress.”
What Do QS Users Want? More!
According to Pew Research Center’s 2013 Internet & American Life Project, 69% of the 3,014 U.S. adults surveyed keep track of at least one health indicator such as weight, diet, exercise routine or symptom. More recently, PwC’s Customer Experience in the Pharmaceutical Sector: Getting Closer to the Patient report found four elements that define the next generation of patients: 1) they want more personalized information and real-time feedback; 2) they are willing to be active participants in their care and treatment; 3) they want more on-demand services that are convenient for their busy lives; 4) they want to be able to value-hunt through comparison shopping. So it appears that the Quantified Self movement will only keep growing.
“Personally, I think it is picking up increasingly because data is becoming so much more accessible and monitoring and self-awareness is picking up, so it is kind of a trifecta,” explains Christopher Means, Executive Director, Sales & Marketing at Digifit, the first app developer to bring heart-rate monitoring to the iPhone, which now offers a suite of apps that track cardio, running, cycling and more. “Also, the barriers to entry are a lot lower nowadays, so it’s no longer just the guy who has maintained an Excel spreadsheet of everything he has eaten since 1992. It’s people who are using mobile apps like MyFitnessPal and then pulling correlations across these different apps and data points.”
However, it is not just people who see the benefit of collecting all of this data. While Chris Bergstrom, Chief Strategy & Commercial Officer at WellDoc, agrees that at its essence the QS movement is about empowering people to better manage their own health, he also understands why the other players in the healthcare ecosystem are encouraged by this movement.
“The ‘establishment’ values the quantified-self movement because it brings access to a data set they have never seen before,” explains Bergstrom. “In fact, it’s the largest data set in all of healthcare—meaning it contains all that occurs to a person in between their lab, physician or pharmacy visits. Further, it is now recognized that only through both empowering patients and also having the visibility to use the ‘real’ patient data, that we can carry out the necessary step of decentralizing healthcare and reversing the course of our unsustainable cost curve.”
The question then becomes how pharmaceutical and medical companies can contribute to this movement and help to improve patient outcomes and/or the healthcare system as a whole.
“Evolutions in technology tend to split the pharma industry into two camps,” says Jen Fuhrman-Kestler, Digital Media Director, Healthcare at GolinHarris. “Those who see endless potential in leveraging technology to help patients lead healthier lives, and those who see insurmountable risk in tackling something unfamiliar.”
To help make the leap into the QS movement a little easier for healthcare companies to understand, PM360 reached out to mHealth and device developers whose products are used by people in the movement, as well as to pharma marketers for their advice, insights and the developments worth keeping an eye on.
Quantified Self mHealth and Device Developers
1. WellDoc’s BlueStar Diabetes Mobile Prescription Therapy (FDA approved for type 2 diabetes)
BlueStar is an FDA cleared, Rx-only product for use by adults with type 2 diabetes that is designed to go “beyond the pill” and move past the traditional focus on data and generic information.
“Through BlueStar’s real-time, personalized education, coaching and patient specific decision support, we can empower patients and their providers to adhere to an optimized treatment plan,” explains Bergstrom. “This improved care coordination and ongoing support helps people living with diabetes face the daily challenges of the disease while helping providers, ACOs and health systems achieve the quality metrics and patient satisfaction scores for which they are now being held accountable. ”
While this is the world’s first Mobile Prescription Therapy, Bergstrom says you should not expect it to be the last.
“Patients have a strong desire to learn about and receive digital health products from their doctor,” explains Bergstrom. “In fact, a recent Digitas Health study showed they have a greater desire for mobile prescriptions than they do for traditional prescriptions. Today, however, doctors can only discuss various digital health products. It will be some time before they have even a handful of true Rx-only Mobile Prescription Therapies available. The hurdles are extremely high from a regulatory, safety, efficacy and end-to-end deployment perspective. So like other prescription products, the time and investment required to bring these products to market is tremendous—although certainly worth the investment.”
Advice to Pharma/Med Device: Bergstrom believes that data will be the currency of healthcare in the future. However, the meaningful use of the information you collect is more important than the data itself. “Medical device and pharma companies must tap into that ecosystem or risk being relegated lower and lower on the value chain. But by moving quickly to partner with and access the leaders in the digital health field, they can collaboratively craft a long-term strategy and sustainable platform for making the transition from widget maker to a broader solutions provider.”
2. OptimizeMe (everyday life tracking app)
OptimizeMe is an app that helps record your life to provide insight into how you can improve it. For instance, you can track your everyday activities, places you visit, people you meet, your mood and more. The app then automatically translates this data into personal insights including: How does walking affect your sleep? How does your work influence your health? How do other people affect your mood?
Bogdan Gerya, Founder and CEO of OptimizeMe, says the app also provides a highly customizable structure that allows the company to tailor it to a broad range of uses including overall patient tracking, patient coaching, drug intake and symptoms tracking, exercise tracking for fitness studios, etc. Gerya adds that while OptimizeMe has not been approached by pharma companies seeking a collaboration, the company has yet to openly communicate their white labeling offering to the general public. In fact, Gerya intends to start approaching a number of pharma companies in Switzerland where the company is based.
Advice to Pharma/Med Device: Gerya says that the Quantified Self movement offers a lot of contextual data, which, if combined with symptomatic data, can help pharma companies to understand significantly more about their patients and could support the ongoing efforts towards more personalized medicine. He also suggests that medtech companies that have long R&D cycles due to strict regulations introduce their products first in the less regulated consumer health and fitness market in order to secure a share in the currently booming wearables market. However, any product meant for the QS audience must:
- Be an intuitive, simple to use solution.
- Provide a clear benefit for the patient.
- Offer open access to own data.
- Provide long-term motivation to keep using the solution.
3. Digifit (mobile cardio fitness solutions)
Digifit offers a suite of apps for iPhone and Android devices that track cardio, running, spinning and other data from ANT+ and Bluetooth compatible sensors, including the Polar H7 BLE Heart Rate Monitor, Scosche Rhythm Heart Rate Monitor and the Fitbit One Activity Monitor. The apps can also sync with already available nutrition mobile solutions such as MyFitnessPal and LoseIt!, because as Means notes, “If you are not paying attention to what you are putting into your body then you’re not going to have much success in weight loss.”
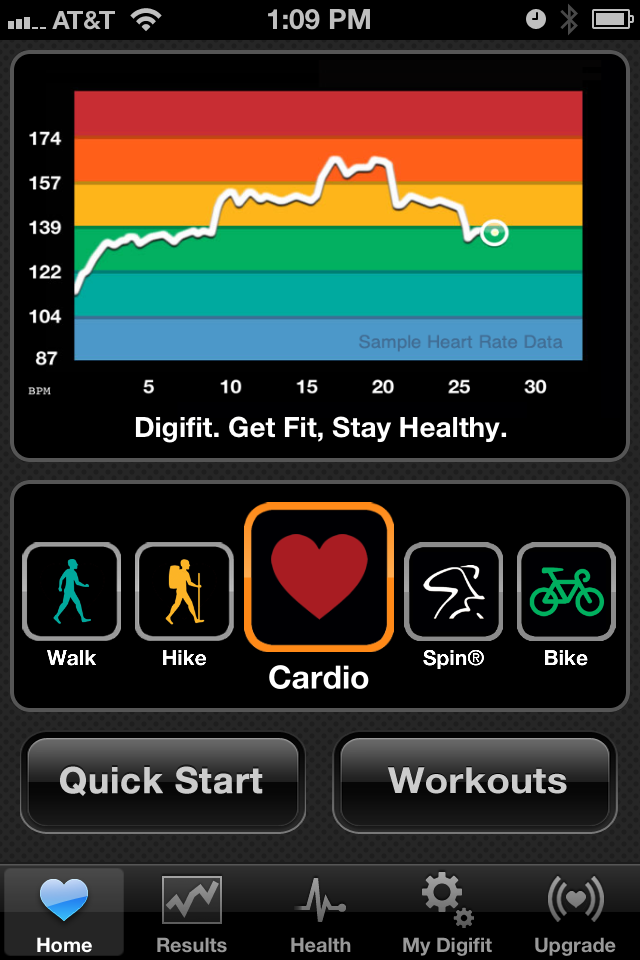
The apps, says Means, also try to understand who the individual is and where they are in their exercise routines and overall fitness levels in order to customize their ideal heart rate zones and exercise programs. But one of the things the company takes particularly pride in: The way their platform displays the data.
“Our goal has been to make the display of data easy and understandable,” explains Means. “I went to CES this year and it had about three different booths there—including one gigantic screen for all of these heart rate monitor and activity monitor companies—and they were displaying our heart rate chart because we spent a lot of time to visualize it, to make it nice, colorful, easy to read, very friendly and informative all at the same time. So you can pretty much get what is going on in the blink of an eye.”
The company now plans to work on more targeted solutions, including some for specific health conditions. He even made it clear that the company would be very willing to partner with a pharma or med device company. One example of the types of more targeted offerings the company is working on includes a solution they developed with Moms in Motion, a group that connects and empowers moms to get fit and hopefully have some fun while doing it. The app includes content they worked on with the NIH to address children’s health and nutrition topics as well as some of Digifit’s training programs.
That example shows that these more targeted solutions don’t all have to revolve around data tracking. Some may be more content heavy, containing educational materials and videos, while others might focus on the social aspect—staying connected with a support group. Means says that the company’s user feedback also reveals that people are looking for more ways to connect and exercise with groups, as well as customized coaching options. And now the Digifit platform can even be used to provide a trainer with access to all of the information for the people in his/her group—and with the ability to offer personalized feedback based on their data. Means says that a similar setup could work for a physician and his/her patients.
Advice to Pharma/Med Device: Means believes that the Quantified Self movement is already hitting the mainstream, so it is probably a mistake for anyone to think this is a passing trend. “The iPhone now has the M7 chip in it that knows when you are running, knows when you are walking and knows your daily activities. That kind of accessibility is increasingly built into phones and more devices,” he explains. “Our particular take on it at this point: The hardware itself is becoming more advanced, less invasive and more publically accessible, so we are focused more on creating experiences that take you from Point A to Point B and provide meaning so that you can make healthy habits a lifestyle choice, rather than something you just do for a period of time.”
4. Tinké (fitness and wellness measuring device)
At the touch of your finger, Tinké can measure your heart rate, respiratory rate, blood oxygen saturation level and heart rate variability. Tinké works via an optical sensor that measures changes in your blood volume—so no finger pricking is involved and the measurement only takes a few seconds. Chua explains that from the four parameters the device measures, it is able to quantify how fit your body is and how stressed you are.
Advice to Pharma/Med Device: Chua says the company is talking with numerous healthcare companies and is happy to see a high level of receptiveness—and a sense of responsibility—from the pharma/medical device companies regarding QS. “The QS movement is revolutionizing the pharma/medical device scene in a series of waves,” she explains. “The sharing of traditional knowledge helps in quickly closing the gap between technology and medical to provide simpler products that translate the benefits to customers/consumers alike, not only for patients but for fit and well individuals who want to maintain/enhance their lifestyle and stay conscientious in the decisions they make to lead better lives.”
5. GERO Lab (researching locomotome science for possible diagnostics)
File this company under one to watch. Right now the research company, based in Moscow, is still conducting various studies as they work towards developing a technology platform for non-invasive diagnostics of chronic diseases in early stages. The company studies locomotor activity, which is a complex technology based on environmental conditions, human reaction to the environment and endogenous stresses. It is proven to be an accurate way of detecting biological markers for human health.
Vera Kozyr, Co-founder and CEO at GERO Lab, offers an easy example of how this works: Think about when you are ill. You immediately notice the change in your behavior. You may lie down more or move a little slower. While these actions are easy to notice, your body is also making tiny, unrecognizable changes as you develop chronic age-related diseases such as type 2 diabetes, Parkinson’s, Alzheimer’s and high-blood pressure.
GERO, however, is able to track these changes by monitoring your precise activity movements through wearables such as those available from Jawbone, Fitbit, BodyMedia and Withings or from the GERO app, which uses the accelerometer in your smartphone. Then, when enough data has been gathered over a long enough period of time—one month with their current algorithms—the company can determine if you are likely to develop any of those diseases.
So far the company has completed a 3,000-user Fitbit study as well as smaller studies with specific diabetes clinics in Moscow. In these studies, they ask for volunteers with one of the diseases they are trying to track in order to see if they are able to accurately diagnose them. Currently, they are accurate 86% of the time for diabetes, 85% for Parkinson’s, 70% for depression and 68% for high blood pressure. Their next goal is to complete a 50,000-user study—and they currently have about 5,000 users signed up, according to Christopher Papazian, Director of Marketing at GERO.
Ultimately, Kozyr says the company wants to develop diagnostic technology that can be approved by the FDA, which means they will need to get their accuracy ratings above the current standard for the diseases they are focused on. While Kozyr says it is possible to detect other diseases, those will have to wait for another day.
Right now, the company is solely focused on the research phase and perfecting accuracy levels as they have yet to really consider their commercialization options. Kozyr does admit that the technology wouldn’t necessarily need to be an app since many clinics and institutions are interested in this functionality. But the company doesn’t even know whether they would apply for approval as they exceed the diagnostic standard for each disease or wait until they reach that standard for all diseases.
In the shorter term, however, the company is considering releasing a product that can determine a person’s Biological Age, which is basically a measure of how well or poorly your body is functioning relative to your chronological age. This wouldn’t require FDA approval and they are getting closer to the desired accuracy level. But the company is also unsure at this time whether this would be something they release as a separate mHealth solution or if they would have it integrated into some other “big player’s” functionalities.
Advice to Pharma/Med Device: Even though the company is some time away from commercializing the technology, Papazian still recognizes some larger trends that are worth paying attention to. “The wearable market needs to evolve past simple metrics for it to have longevity,” he explains. “As the Quantified Self movement is improving, increasing numbers of people are going to want these things—but not the invasiveness of implants.” The problem right now is that most people only wear their Fitbit, Jawbone Up or similar device for a few months before ditching it as the data doesn’t mean much to them. Or as he puts it, “Great I did 12,000 steps. Does that mean I can have three beers and some chicken wings now?” The key will be to deliver information that is valuable to users, such as where their health is headed.
Advice for Entering the Wearables Market
Wearables are obviously a large part of the QS movement and pharma companies seem intrigued by the possibilities. Consider these six tips if your company is thinking about entering this market.
1. Pay Attention to Apple: Jennifer Hague, Manager, Social Media at Intouch Solutions notes that, according to SiliconAngle.com, Apple has been granted a patent for a biometric headphone system that can detect various metrics, such as temperature, heart rate and perspiration levels. But, of course, Hague says Intouch is not putting all of its apples into one basket as the agency has several studies underway that are exploring how apps can benefit a patient’s life. This includes a trial app that shows the benefits of mobile devices and connectivity as medicine, delivering biofeedback to an online health platform.
2. Solve Unmet Needs: Just as pharma companies look to develop drugs to solve unmet patients’ needs, manufacturers need to deliver the right technology to solve unmet needs in the wearables market, explains Fuhrman-Kestler from GolinHarris. This may require producing or acquiring an original piece of wearable technology, or developing an app for existing wearables such as Google Glass or Samsung Galaxy Gear. She adds, “Regardless of the hardware, a fundamental contributor to the success of a healthcare wearable is its ability to drive and reward positive behavioral change by enabling smart goal-setting in a highly personalized, life-simplifying and sustainable way.”
3. Comfortable and Attractive Designs: Fuhrman-Kestler also believes that even though designing for aesthetic appeal is mostly unchartered territory in this industry, embracing design challenges also presents new opportunities. For instance, a conversation-starting wearable could appeal to patient populations looking to raise awareness of their disease state. However, other patient populations may prefer to “blend in” and would not want yet another physical reminder of their disease. From that perspective, it would be best to develop an app for an existing device such as Google Glass or Samsung Galaxy Gear.
4. Provide Real Health Value: Geoff McCleary, VP, Group Director, Mobile Innovation at Digitas Health, believes that the challenge for pharma (and the broader health industry) will be to put real health value to this data in a meaningful way. While these devices are getting good at counting steps, calories burned and basic bio functions such as heart rate, McCleary thinks the products need to take this a step further to start answering questions such as: How do my heart rate and breathing impact how I metabolize medicine? How is my medication compliance affected by my daily caffeine intake? Why will I get better outcomes if I eat a more protein rich diet in the morning? He also thinks we could benefit from a standard universal health profile that holds all of our health data: Past (doctor visits and test results), Present (device data and medication activity) and Future (DNA profiles and risk).
5. Deliver Outcomes: Bernhard Kappe, Founder and President of Pathfinder Software, a company that helps medical device and healthcare companies design, develop and launch the right software and mobile apps, believes that before a pharma company decides to contribute to this movement it has to determine the value of engaging with QS users. He notes that current business models are undergoing disruption as payers and providers are making purchase decisions and determining pricing more on the basis of real world outcomes rather than point solutions recommended by physicians. Put simply: “If you have systems that improve outcomes, you create value and you can get paid for that. If you don’t, you risk being commoditized (the way test strip manufacturers in the diabetes space are).”
But Kappe does have one example that pharma can learn from. A company called Propeller Health develops sensors that attach to inhalers for Asthma and COPD. The sensors send signals—via Bluetooth through a mobile phone—to the cloud whenever the inhaler is used. This data, along with data from other users as well as environmental data, is processed and used to provide recommendations to help patients manage their condition. Kappe explains that this results in outcomes that are equivalent to, or better than the best asthma medication, and provide a $600 per patient per year cost reduction for the payer.
6. Imagine the Future: Greg Cohen, Sr. Manager, Social Media & Influence at UCB, pictures a day when wearable devices can deliver an experience that is very similar to what GERO is working on. “There should be a future in which the Quantified Self movement goes from counting steps and monitoring sleep to potentially identifying certain diseases in early stages,” he says. “A worn device could measure slight changes in gait, imperceptible tremors and elevated heart rates—all potential early indicators of certain diseases.” But Cohen also sees the benefits that this brings to the patient-physician relationship.
“Instead of the usual ‘I’m feeling fine,’ a patient could actually have a quantitative report showing exactly what’s going on,” explains Cohen. “Certain appointments might even be avoided completely depending on the level of sophistication of the product, clearing the physicians’ schedules for patients with greater immediate need.”
Cohen also suggests developing a wearable that monitors pain levels that can indicate when patients and physicians need to meet—or even an algorithm that could compile symptoms/vitals to cross-reference with current patient meds. Then the result could present possible pharmaceutical solutions for the physician to review with the patient based on that patient’s personal situation.
“When properly applied, the Quantified Self movement is beneficial for patients, HCPs and the pharma industry as a whole,” says Hague. “It is a positive development that encourages a healthy lifestyle, holds a patient accountable for his or her decisions, increases disease awareness and improves adherence to prescribed treatments.”