Medical Device: A “Smart” Cane in Development Enables the Visually Impaired to Recognize Familiar Faces in an Instant
A revolutionary “smart” cane that enables the visually impaired to instantly identify friends and family could soon be available, thanks to students at Birmingham City University. The XploR mobility cane uses smartphone GPS functionality technology to recognize familiar faces and aid navigation. It is designed to detect faces up to 32 feet away and vibrate when detecting a recognizable individual from a bank of images stored on an internal SD memory card. The Birmingham City University team presented the XploR cane to medical and science professionals in Luxembourg and France, and it plans to visit organizations in Germany later this year.
Doctor Docs: Hashtags Enable Cancer-Specific Information
A poster displaying a new series of Twitter hashtags dedicated to different types of cancer was revealed at this year’s American Society of Clinical oncologist meeting. Dr. Matthew Katz, a radiation oncologist at the University of Texas’ MD Anderson Center, stated, “Our hypothesis is that cancer-specific hashtags are a promising way to facilitate access to accurate health information and positive interactions. Our aim is to retrospectively evaluate Twitter use of the Cancer Tag Ontology and the types of users putting these hashtags into tweets.”
The goal of the cancer-specific hashtags is to help stimulate and direct Twitter conversations to provide another source of information, boost patient education and stimulate advocacy.
TeleMed Texts: Steps For Improving EHRs
Electronic health records should support person-centric care delivery by 2020, according to an American Medical Informatics Association panel. To ensure this, health IT developers and policy makers should work on: Simplifying and increasing the speed of clinical documentation; refocusing government regulations; increasing transparency of EHR certification; and promoting innovation, according to the AMIA EHR-2020 Task Force. The Task Force issued 10 recommendations in those four areas:
- Lessen the data entry burden for clinicians.
- Separate data entry from data reporting.
- EHRs should support systematic learning and research at the point of care during routine practice for both billing and care-delivery purposes.
- Regulators should clarify and simplify EHR certification procedures; emphasize health information exchange and interoperability; cut the need for duplicate data entry; and make patient outcomes the goal of certification, rather than EHR functionality.
- Reimbursement rules should “support novel changes and innovation in EHR systems.”
- More transparency is a must for the EHR certification process to improve usability and patient safety.
- Healthcare providers and vendors alike “should be fully transparent about unintended consequences and new safety risks introduced by health information technology systems, including EHRs, as well as best practices for mitigating these risks.”
- Vendors should use application-programming interfaces and follow public open data standards.
- EHRs should be integrated into the “full social context of care, moving beyond acute care and clinic settings to include all areas of care: Home health, specialist care, laboratory, pharmacy, population health, long-term care and physical and behavioral therapies.”
- User interfaces should be intuitive because, as the article notes, “Usability is a real science and goes beyond screen design.”
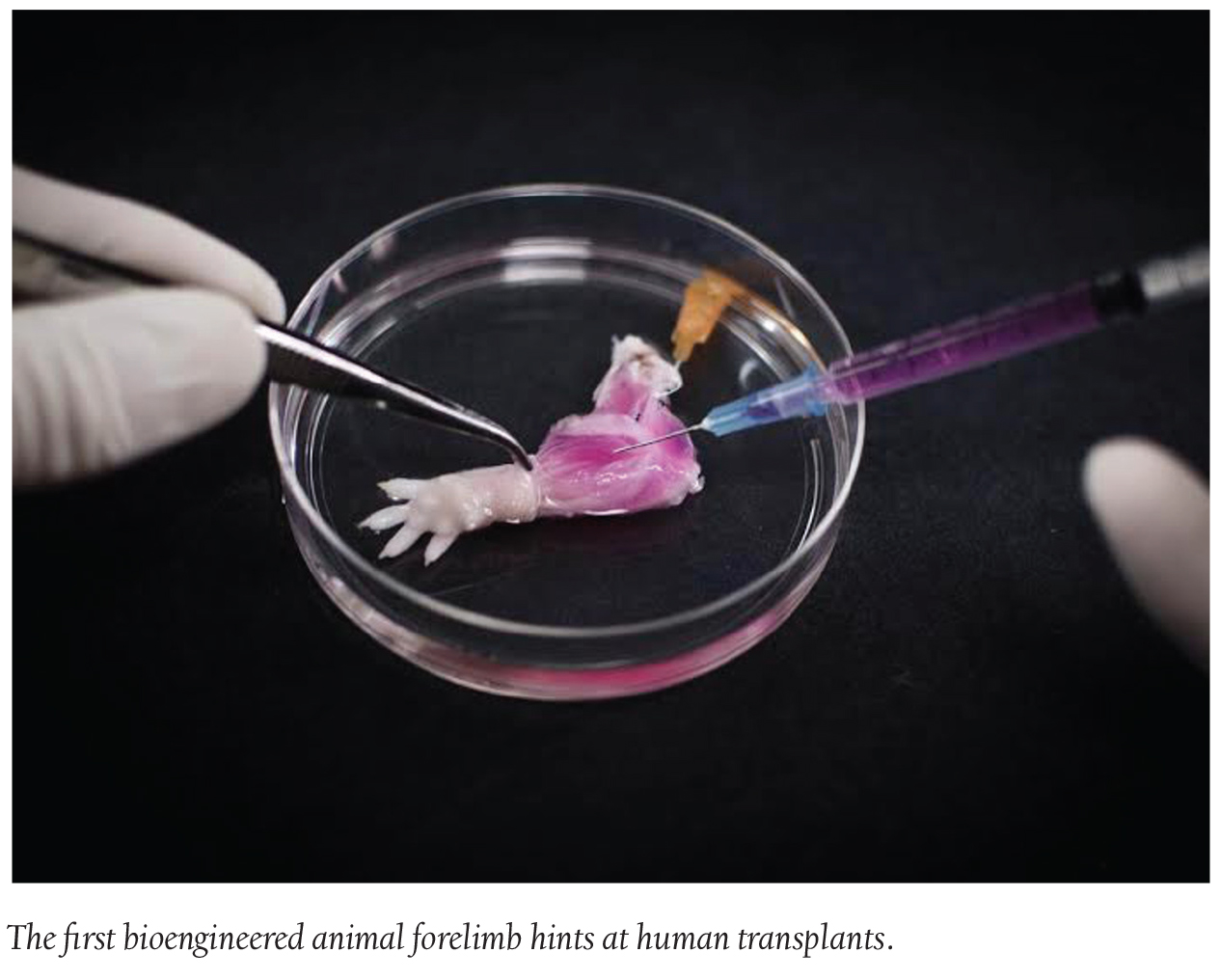
Discoveries/Innovations: First Bioengineered Forelimb Developed
The first steps towards developing bio-artificial replacement limbs suitable for transplantation were taken by a team of Massachusetts General Hospital investigators. In their report, the researchers describe using an experimental approach previously used to build bio-artificial organs to engineer animal forelimbs with functioning vascular and muscle tissue. This is the first reported use to engineer the more complex tissues of a bio-artificial limb.
Sales Sector: Cancer Drug Payment Deals Sought
Express Scripts Holding Co., a large manager of prescription-drug benefits for U.S. employers and insurers, is seeking indication-specific pricing for prescription drugs. Currently, Express Scripts and most insurers pay the same per-unit rate for a cancer drug regardless of the type of cancer it is being used to treat.
“One of the big frustrations has always been people paying top dollar for drugs that aren’t always giving them the best response,” Express Scripts Chief Medical Officer Steve Miller told the Wall Street Journal. “If pharma is truly sincere about wanting value-based reimbursement, we now have the sophistication to do that.”
Dr. Miller pointed to Tarceva, a drug co-marketed by Roche Holding AG and Astellas Pharma Inc., which has shown a smaller benefit in pancreatic cancer than in lung cancer. In an “indication specific pricing” model, the per-pill cost of Tarceva would be lower for pancreatic-cancer patients than for lung cancer patients, given the reduced efficacy, the doctor notes. The company has begun approaching drug makers about arranging such deals, which could go into effect for 2016.
FDA Update
ED Drugs Not Linked to Melanoma
A causal link between the use of erectile dysfunction drugs—Pfizer’s Viagra, Lilly’s Cialis and Bayer and GSK’s Levitra—and greater risk of contracting malignant melonoma has not been found, according to a study recently published by JAMA.
While 4,065 men were diagnosed with the illness between 2006 and 2012, only 435 were found to have developed the skin cancer, according to the report. Instead JAMA suggested that among other factors such as certain lifestyles that afford a higher income, more education and sun exposure due to frequent vacations might put more men at risk—but more research needs to be done.
FDA Approvals
The injectable therapy used as an adjunctive to percutaneous coronary intervention, The Medicines Company’s, Kengreal, was approved by the FDA recently for adults who have not been treated with a glycoprotein 11b/111a or a P2Y12 inhibitor.
FDA Approval Recommendations
Repatha, a cholesterol-lowering drug developed by Amgen was recently recommended for clearance by an FDA advisory committee. The drug will be used in patients with hypercholesterolaemia. Amgen will receive a final decision on August 27.
Sanofi and Regeneron’s Praluent, was backed by a FDA panel for approval to reduce LDL levels and reduce cardiovascular event risks.
The FDA also granted an extended review to Allergan and Gedeon Richter for cariprazine, an atypical antipsychotic for treatment of schizophrenia and acute bipolar 1 disorder manic or mixed episodes, until September.