Doctor Docs: How HCPs Want to Engage with Pharma Now

Since the pandemic, pharma has worked to adapt to the new needs of HCPs. But, according to new research from Accenture, pharma companies still have work to do to better understand and meet the expectations of HCPs and the needs of their patients. In fact, HCPs today still feel as though pharma companies are failing to understand the real impact of COVID-19 on them (56%) and their patients (60%, up 9% from 2020). Meanwhile, 65% of those surveyed felt that a pharma company has “spammed” them with digital information, indicating that brands need to tune into what information is truly helpful to keep important information from being lost in an inundation of communications.
Importantly, the survey found that pharma companies who provide more meaningful engagements are rewarded with more time and attention. In fact, 88% of HCPs would be twice as likely to meet with other companies and representatives if they replicated the best relationship they have formed. And what do HCPs value from these relationships? Content that helps them better serve their patients. Half of what HCPs consider the “best content” they received was either content they could easily pass to their patients for support or content helping them with the administration associated with treating their patients.
While virtual meetings are here to stay, almost half of the HCPs surveyed said they prefer a mix of virtual and live engagement. This is important to keep in mind as 44% of HCPs said discussions with pharma representatives have the most influence on their treatment decisions. Pharma companies need to keep up the trend of delivering more meaningful content rather than inundating HCPs with more content to receive more attention and better engagement, especially when it comes to patient care during the COVID-19 pandemic.
Trendsetter: Walmart Launches New Shop-By-Diet Tool
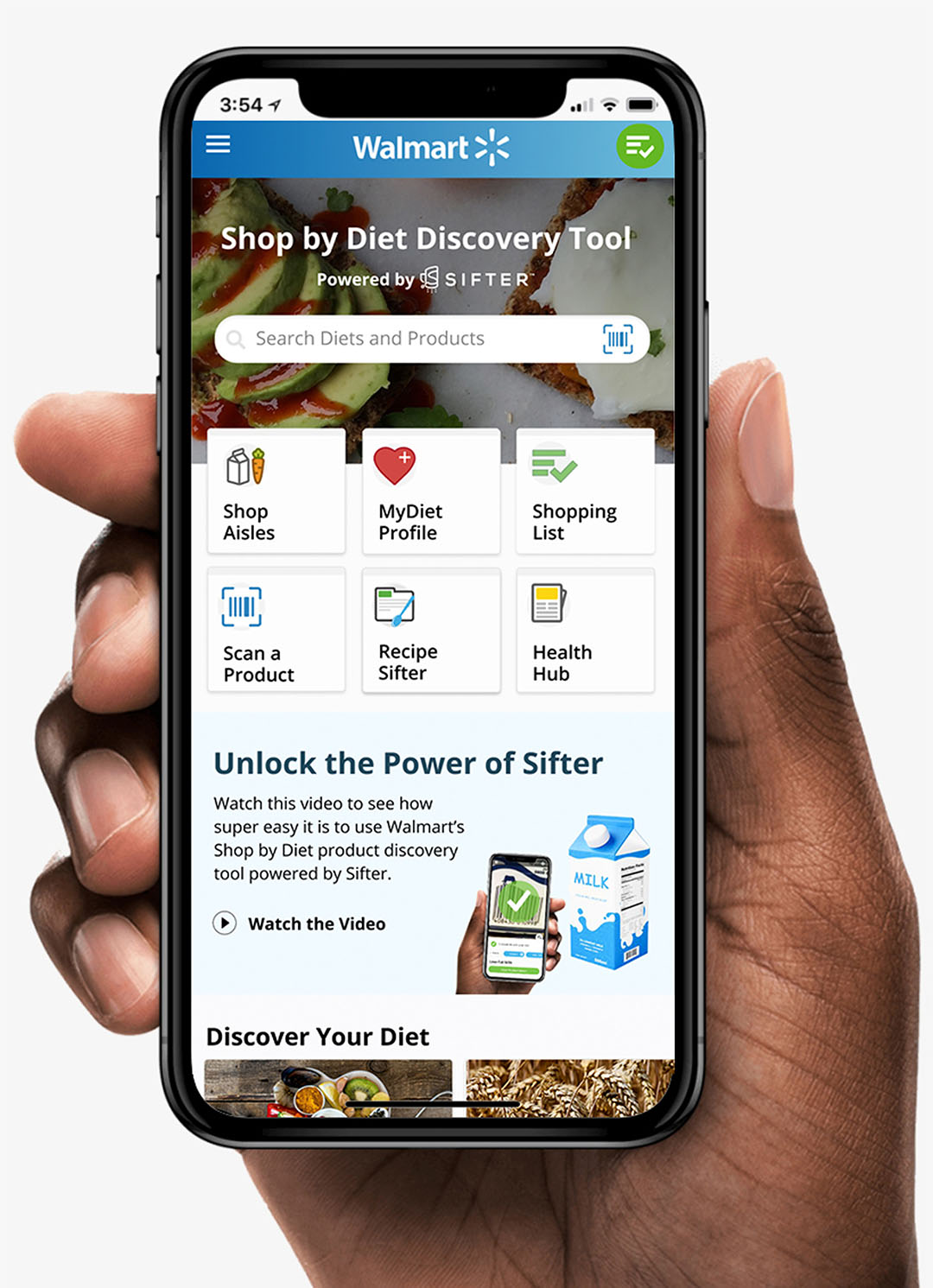
Walmart is helping shoppers take control of their health with a new approach to online and in-store grocery shopping. The company signed a deal with Sifter SP Inc. to use their Nutrition-as-a-Service platform to build a shop-by-diet tool for Walmart consumers, more than 200 million of which are reportedly adhering to a diet or health-related program, while 85 million are conscious of food allergies or avoidances when shopping.
The in-store tool will allow customers to input preferences, such as “no dairy” or “no gluten,” and scan any item in the store with the Walmart app on their smartphones to see if it matches with their priorities. The online tool helps shoppers filter grocery items with the same such parameters, enabling them to discover and shop for foods based on allergens; medical diets such as diabetes or heart health; popular lifestyle diets such as vegan, keto, or kosher; and other options that include grass-fed products or products that won’t interact with medications.
“Partnering with Walmart to support customer wellness is exciting for everyone at Sifter,” said Andrew Parkinson, Co-founder of Sifter. “Sifter’s passion is to make healthy eating easy for everyone. Walmart’s Shop-by-Diet tool will help millions of its customers better manage their health through diet and nutrition.”
The service rollout is the latest in Walmart’s efforts to help customers and patients take control of their health and reach their lifestyle goals as a go-to health partner to their everyday consumers.
Patient Pages: Is the COVID Vaccine Really Free in the U.S.?

As the U.S. government mobilized major COVID-19 vaccine programs, citizens were promised free shots and tests across advertisements and media, regardless of insurance status. However, a new survey by AffordableHealthInsurance.com found that 50% of Americans paid for at least one COVID-19 test administered at a health clinic, government site, or pharmacy, while 42% of vaccinated Americans paid for their shots. Of the 42% billed patients, only 57% were aware they’d be charged. Adding to the uncertainty, 35% of those who knew they would be charged did not know how much they would be charged before receiving their bill.
While the CDC site claims that the vaccine is available free of charge to all those 5 years of age and up, of the 1,250 American adults surveyed, 32% of uninsured Americans got their vaccine for free compared to 66% of insured Americans. Surprise bills or not, it’s apparent Americans don’t trust the word of the government and it may be affecting their willingness to get vaccinated. When the survey asked about the validity of the statement, “COVID-19 vaccines are free to all people living in the United States, regardless of their immigration or health insurance status,” 26% said it was false. Meanwhile, 44% of Americans who don’t believe the vaccine is free remain unvaccinated against COVID-19 while 31% of Americans who do believe the CDC’s statement about free vaccines aren’t vaccinated.
Unfortunately, when it came to treatment, 28% of Americans avoided COVID-19 treatment because they feared the cost and surprise bills. And in fact, of the 30% of those surveyed who sought treatment for COVID-19, 70% were charged—42% unknowingly. Again, those who knew they would be charged, did not know how much the cost would be until they received their bill (37%). Health insurance offers little relief in these uncertain situations, since insured Americans were only slightly more likely to receive COVID-19 treatment free of charge (33% vs. 26%), adding to the reluctance many feel to be treated for the potentially fatal disease during the pandemic.
FDA Update
Drug Approvals
The FDA issued emergency use authorization to Merck’s molnupiravir for the treatment of mild-to-moderate coronavirus in certain adults. Patients with positive results of direct SARS-CoV-2 viral testing who are at high risk of disease progression may be candidates for the drug. Molnupiravir is prescribed to those at risk of hospitalization or death and intended to be given no later than five days after the onset of symptoms.
The Leqvio (inclisiran) injection has been authorized by the FDA as a supplemental treatment for adults with heterozygous familial hypercholesterolemia (HeFH) or clinical atherosclerotic cardiovascular disease (ASCVD). Novartis’ drug is to be used along with a special diet and maximally tolerated statin therapy and is intended to reduce circulating levels of LDL-C.
The FDA approved a new medication for myasthenia gravis (gMG), a chronic autoimmune, neuromuscular disease that causes weakness in the skeletal (voluntary) muscles that worsens after periods of activity. In gMG, the immune system produces AChR antibodies that interfere with communication between nerves and muscles, bringing on weakness attacks that can cause potentially fatal breathing and swallowing problems. Vyvgart (efgartigimod) can be prescribed to adults who test positive for these antibodies. argenx BV developed Vyvgart as a new class of medications, a fragment of antibody that binds to receptors on cells that cause an overall reduction in AChR antibodies.
Med Device Approvals
The FDA granted premarket approval to OrganOx’s metra system, a fully automated normothermic machine perfusion (NMP) device for the preservation and transport of donor livers destined for transplantation. The marketing approval includes both livers from donors after brain death (DBD) and livers from donors after circulatory death (DCD). The device is capable of sustaining donor livers destined for transplantation in a functioning state for a total preservation time of up to 12 hours.








