Med Device: A New Wearable Can Help Prevent Pneumonia
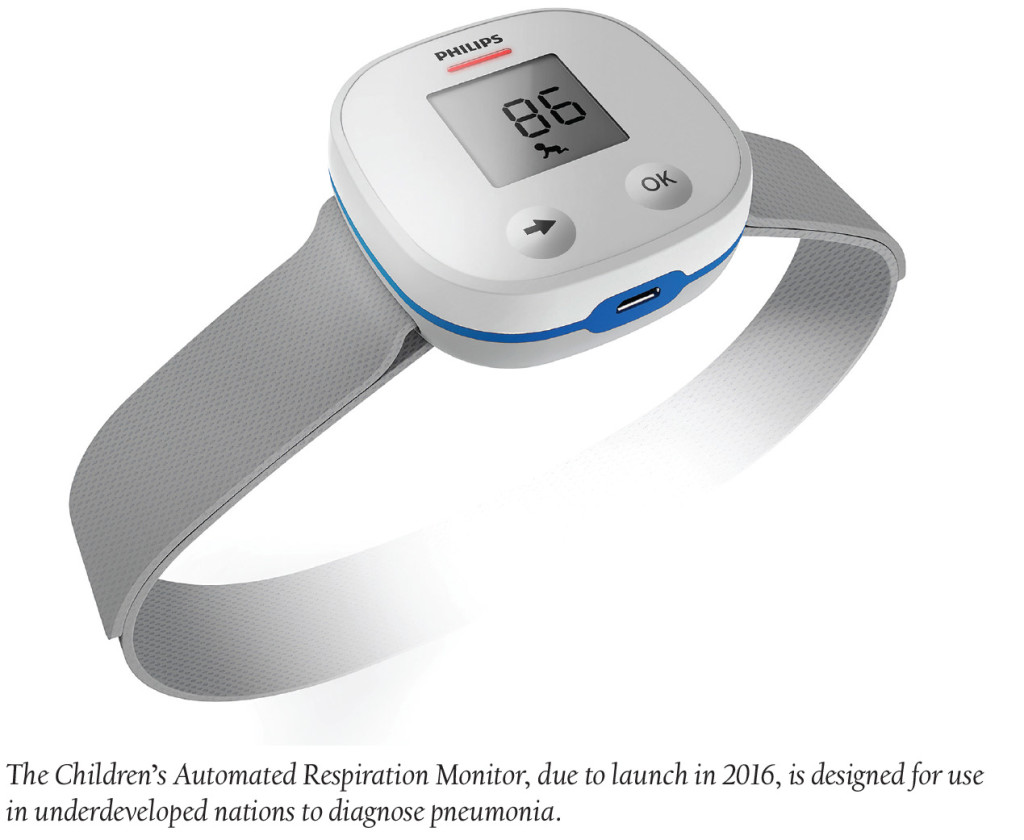 Medical devices to help stave off pneumonia in poorer countries, the most common killer of children globally, have been produced by multiple companies since 2011. But a new device, designed by Royal Philips, the Children’s Automated Respiration Monitor, is on track to launch in 2016. The device will count breaths and the speed of respiration and send accelerometer signals to a wrist-band monitor, giving HCPs critical information for diagnosing pneumonia. Currently, healthcare workers in these underdeveloped nations attempt to conduct visual breath inspections.
Medical devices to help stave off pneumonia in poorer countries, the most common killer of children globally, have been produced by multiple companies since 2011. But a new device, designed by Royal Philips, the Children’s Automated Respiration Monitor, is on track to launch in 2016. The device will count breaths and the speed of respiration and send accelerometer signals to a wrist-band monitor, giving HCPs critical information for diagnosing pneumonia. Currently, healthcare workers in these underdeveloped nations attempt to conduct visual breath inspections.
CEO of Philips Africa, J.J. van Dongen, says, “Philips’ vision is to improve lives through meaningful innovation. The population growth is highest in emerging markets including Africa and South East Asia, and innovation can help drive sustainable solutions that bridge the divide between the privileged and less privileged sections of society to improve the quality of life at all levels.” While the monitor will help healthcare workers prescribe antibiotics and treat pneumonia before it is too late for many children, it cannot detect the underlying cause of illness. Philips’ device will be available in countries including Kenya, the Netherlands and India.
Therapeutic Talk: Gene Therapy Moves Closer to Curing Batten Disease
Batten disease is a deadly, inherited illness that blocks the ability of children’s bodies to dispose of cellular waste. But researchers from Dr. Beverly Davidson’s team at the Raymond G. Perelman Center for Cellular and Molecular Therapeutics at The Children’s Hospital of Philadelphia recently discovered that dogs with a similar disease tend to live longer when they possess a crucial enzyme that is lacking in Batten disease patients. Work is now being done with gene therapy to produce this enzyme, which will allow the brain to then signal the body to dispose of waste.
The enzyme, researchers note, would be injected into the brain. Similarly affected dogs were injected with a virus containing the gene needed to develop the enzyme, which spread through their brains and resulted in dramatic clinical improvement. Although the dogs were not cured, the development of the disease was slowed and the dogs were then better able to walk and interact with other dogs. According to Dr. Davidson, “If the outcome is equally profound in children with the same enzyme deficiency, this would represent a great benefit for affected children and their families.”
Discoveries/Innovations: Nestlé To Combine Food and Medications To Treat Illnesses
A health-focused unit at Nestlé has been hard at work over the past four years planning a fusion of food and medicine to treat a range of health conditions. The team, led by CEO Greg Behar, is discussing approval processes with the FDA and targeting highly variable health markets such as those for Alzheimer’s and stomach diseases. Last month, the company partnered with AC Immune to begin research for the early detection of Alzheimer’s. Nestlé also backed Seres Therapeutics with $65 million to develop methods to restore the microbiomes that keep the stomach healthy.
To develop a research hub in New Jersey, Nestlé will invest $70 million, a cost the company can easily afford given their current annual revenue of $2 billion. In a statement to Reuters, Behar said, “The FDA wants to bring innovation to the market. They see us as a partner that can help shape this area and this regulatory framework.” The CEO is very confident about the company’s endeavors and expects to gain $10.5 billion annually through its innovations.