Oncology drug and biologics makers face increasing demands for data that demonstrate the benefit and cost-effectiveness of their products. Not only are treatment cost increases considered unsustainable, but questions about new product effectiveness are also openly raised by physicians and payers alike. Pharmaceutical companies are increasingly expected to demonstrate the overall economic and clinical value (ECV) of new products to justify premium payment.
The End of “Special Status?”
Historically, cancer drugs have enjoyed “special status” because they are used to treat patients with generally incurable diseases whose life expectancies are often measured in months. This special status permeates clinical medicine where new drugs and biologics are rapidly adopted, even when the scientific rationale is weak. It has also permeated regulatory approval processes, marketing authorization and reimbursement policies. In fact, the cost and value of new cancer treatments has (historically) rarely been questioned. Payers have typically deferred to oncologists in their network when establishing coverage and reimbursement policy. Policy and common practice have traditionally provided mostly unlimited access to drugs and biologics since most of these products would only be used for very short periods of time—often only providing marginal benefits.
But the world is changing. As a result of an aging population, significant therapeutic innovation and increasing patient survivorship, the cost associated with cancer treatment has skyrocketed over the past two decades, and the cost of cancer drugs is expected to continue growing at more than 20% annually over the next few years.1 New products and increased spending on treatments, however, have not always resulted in significant improvement in patient survival rates.2, 3 As a result, payers are requiring data on new drugs’ cost-effectiveness—not just safety and efficacy—before agreeing to reimburse them.
Stakeholders Debate Value of New Treatments
While many new oncology treatments are truly changing cancer survival for the better, new oncology products are not immune to the growing global focus on overall economic and clinical value. The advent of new and more expensive treatments has led a range of stakeholders (patients, clinicians, payers, policy makers and society) to join the debate on the value of new treatments in oncology. Demonstrating economic and clinical value to each of these stakeholders will be critical and represents the next step in the evolution and maturation of evidence-based medicine (EBM). The focus on economic and clinical value has important implications for the future of successful drug and biologic development and innovation in oncology.
Demonstrating ECV is, however, about more than just managing drug costs due to constraints on healthcare spending. The emergence of the value discussion in oncology is part of a broadening debate around the quality of cancer care, access to treatments and changing stakeholder expectations. The value of any treatment is already being examined in the context of HTAs, predictive care paths, bundled payments and certain types of insurance policies. Increasingly, new treatment coverage decisions are based on the demonstration of measurable value.
Controlling Costs Globally
Approaches to controlling cost vary from country to country, however, cost effectiveness is increasingly important. For example, in a multi-country study (including Australia, Canada, England, France, Italy, Germany, Japan, New Zealand, the Netherlands, Scotland, Sweden, and the U.S.) evaluating variability in cancer drug reimbursement found:
- Finland, France, Germany, Sweden, and the U.S. reimbursed the most indications (range: 90%–100% of possible indications).
- Canada (54%), Australia (46%), Scotland (40%), England (38%), and New Zealand (25%) reimbursed the fewest.
All five countries incorporated a cost-effectiveness analysis into reimbursement decisions. Rejected reimbursement submissions mainly occurred because of lack of demonstrated cost effectiveness.4
But demonstrating the overall value of a new oncologic agent is particularly challenging. Cancer treatment is complex, and new drugs may only have incremental clinical benefit for a subset of the patient population. Moreover, it can be difficult or impossible to accurately predict a drug’s effectiveness for a given patient in a “real world” scenario before it is administered.
The Rise of Risk Sharing Agreements (RSAs)
That’s why national health authorities are increasingly “hedging” their reimbursement decisions with RSAs, pricing controls or negotiated price cuts to control costs. RSAs in particular are increasing in frequency around the world.
The best known among these include Pfizer’s Sutent (sunitinib malate) and Onyx/Bayer’s Nexavar (sorafenib) anti-cancer drugs. The concept is similar to a money back guarantee; RSAs provide an assurance of quality and confidence. Some consider risk-sharing (also known as outcomes-based contracting or performance-based agreements) as simply a way to cut costs. In the U.K., for instance, risk-sharing deals have helped the National Institute for Health and Clinical Excellence (NICE) reduce cost per patient.
In the U.S., on the other hand, drug coverage and reimbursement for approved oncology indications is virtually assured. Medicare is required to cover all on-label indications, and has limited ability to control pricing for patented drugs. Additionally, Medicare covers off-label applications included in various compendia. Although the laws vary by state, commercial payers often have more flexibility to restrict access in the off-label setting by requiring preauthorization or large co-pays for high-priced drugs.
The FDA also offers a facilitated review process for drugs based on unmet clinical needs. Novartis’ Gleevec (imatinib mesylate), was approved in just two and a half months as a result of the accelerated approval process, and Millennium Pharmaceuticals’ Velcade (bortezomib) went through the accelerated process even quicker—only two months.
The Changing U.S. Reimbursement Landscape
Increasingly, however, even in the U.S., reimbursement rates for oncology drugs are contingent upon evidence of improved patient survival rates. For example, Centers of Medicare and Medicaid Services (CMS) decided to reject AstraZeneca’s Iressa (gefitinib) from Medicare Part D formularies because it showed no impact on survival among non-small cell lung cancer (NSCLC) patients.
Additionally, providers in the U.S. are focusing on costs as well. In one recent case, physicians at Memorial Sloan-Kettering Cancer Center decided not to use Zaltrap, a new drug for treating metastatic colorectal cancer. The available data indicated that Zaltrap is no better at extending life than the product currently used (Avastin) and it costs twice as much ($11,063 on average for a month of treatment vs. approximately $5,000).
Cancer researchers are also focused on the issue of cost-effectiveness. For example, at the 2012 American Society of Clinical Oncology (ASCO) annual meeting, 44 studies examined cost-effectiveness issues—about twice as many studies as in 2005.5 This focus on cost-effectiveness means that companies must develop and demonstrate that their products provide a meaningful benefit for patients compared to existing treatments and products.
Finally, professional societies are increasingly joining the debate on the value of new and existing treatments in oncology. Since 2012, ASCO has been publishing recommendations as part of its “Choose Wisely” initiative, intended to improve quality and reduce costs by eliminating overuse of specific tests and therapies.6,7 While these are offered as recommendations now, it’s likely that they will be used as a rationale by some payers for limiting or denying reimbursement for products and services at some future point.
Overcoming Barriers in Oncology
Companies with a strategic focus on oncology must accept the fact that the market has changed, with many treatments established and payers using new criteria to evaluate expensive novel products. While payers, providers and researchers all offer increasing scrutiny of expensive cancer treatments, this space continues to present enormous opportunity as our understanding of cancer, focus on personalized medicine, biomarkers and development of more targeted diagnostics all increase.
For pharmaceutical companies, these trends may drive a different approach to product development and commercialization, one that includes a greater focus on economic and comparative data tailored to meet the needs of a more diverse set of stakeholders. This approach includes a realistic understanding of what payers in different countries are prepared to pay for and the ability to meet their criteria and maximize the possible access across countries.
References:
1. “Drug Costs Now A Bigger Issue In Cancer Treatment,” By Deena Beasley, updated 6/15/2012. http://www.msnbc.msn.com/id/47829069/ns/health-cancer/#.T-OTHJFxuHQ.
2. “Cancer Survival Rates for Selected Cancer Sites by Sex and Race,” Centers for Disease Control (2009).
3. http://seer.cancer.gov/faststats/selections.php?series=cancer, Surveillance Epidemiology and End Results (2013).
4. “International Variability in the Reimbursement of Cancer Drugs by Publically Funded Drug Programs,” Current Oncology, P.K. Cheema, et. al., 2012 Jun;19(3):e165-76. doi: 10.3747/co.19.946.
5. “Drug Costs Now A Bigger Issue In Cancer Treatment,” By Deena Beasley, updated 6/15/2012. http://www.msnbc.msn.com/id/47829069/ns/health-cancer/#.T-OTHJFxuHQ.
6. L.E Schnipper et al. “American Society of Clinical Oncology Identifies Five Key Opportunities to Improve Care and Reduce Costs: The Top Five List for Oncology.” Journal of Clinical Oncology. 2012 30(14): 1715-24.
7. L.E. Schnipper et al. “American Society of Clinical Oncology 2013 Top Five List in Oncology.” Journal of Clinical Oncology. doi:10.1200/JCO.2013.53.3943.
Sidebar: How Oncology Drug Makers Can Meet Increased Payer Demands
As new oncology products are developed, ensuring that economic and clinical value considerations relevant to payers and other stakeholders are taken into account is critical. The challenge that oncology companies face is that perspectives on product value can differ. Payers will generally view the value of a new product in terms of its potential to improve patient management (i.e., reduce side effects and ED visits) and overall treatment (medical) costs, but the same data may be viewed differently in different countries.
Evaluate New Products Based on Potential
Oncology drug makers will need to examine new products’ potential carefully and look beyond traditional patient populations—which, in the eyes of payers, may already have numerous treatment options. The more treatment options available (Figure 1), the greater the need to demonstrate significant improvement in clinical outcomes (e.g., differentiated value in overall survival, fewer side effects) and cost-effectiveness compared to current standard of care. Ensuring market access requires fresh thinking about comparative effectiveness, competitive set, the collection of economic and clinical value data, and using such data to position products for reimbursement.
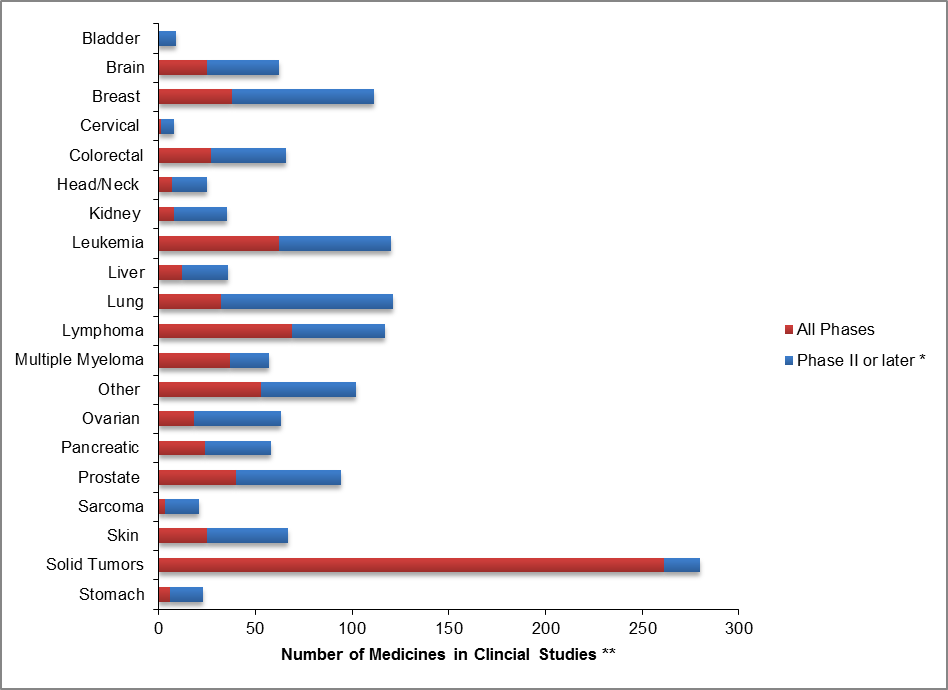
Source: PhRMA 2012 report
*Includes medicines with the following development status: phase II, phase II / III, phase II completed, phase III, and phase III completed
** Some medicines are listed in more than one category
For comparative purposes, when evaluating new products the focus must be on the current standard of care and existing evidence-based care paths such as those supported by the National Comprehensive Cancer Network (NCCN). For example, determining the potential of a new drug to take the place of multiple drugs in a current therapeutic regimen would be important. In that case, demonstrating equivalent efficacy (vs. superiority) may be sufficient. Other areas of interest to both providers and payers include lower dosing frequency (and improved patient compliance), fewer side effects or fewer physician visits.
All of these issues will require difficult decisions about clinical endpoints to pursue and specific indications to target. Companies must examine whether data for a smaller patient population will enable the development of a stronger value proposition—which may raise the potential for premium pricing and enhanced market access.
Given the significant market differences, companies must prepare dossiers that address specific country requirements and perspectives. The same product won’t get the same reception across Europe, and U.S. payers are also demanding more. Companies must also be prepared for the increasing use of risk-sharing agreements.
To meet these demands, market access professionals must establish and maintain close contact with payers, government agencies and relevant professional and patient organizations, soliciting and using their input across the product development and commercialization process. Equally important, developing trust and credibility serves as an important base on which to build the case for both approval and reimbursement.
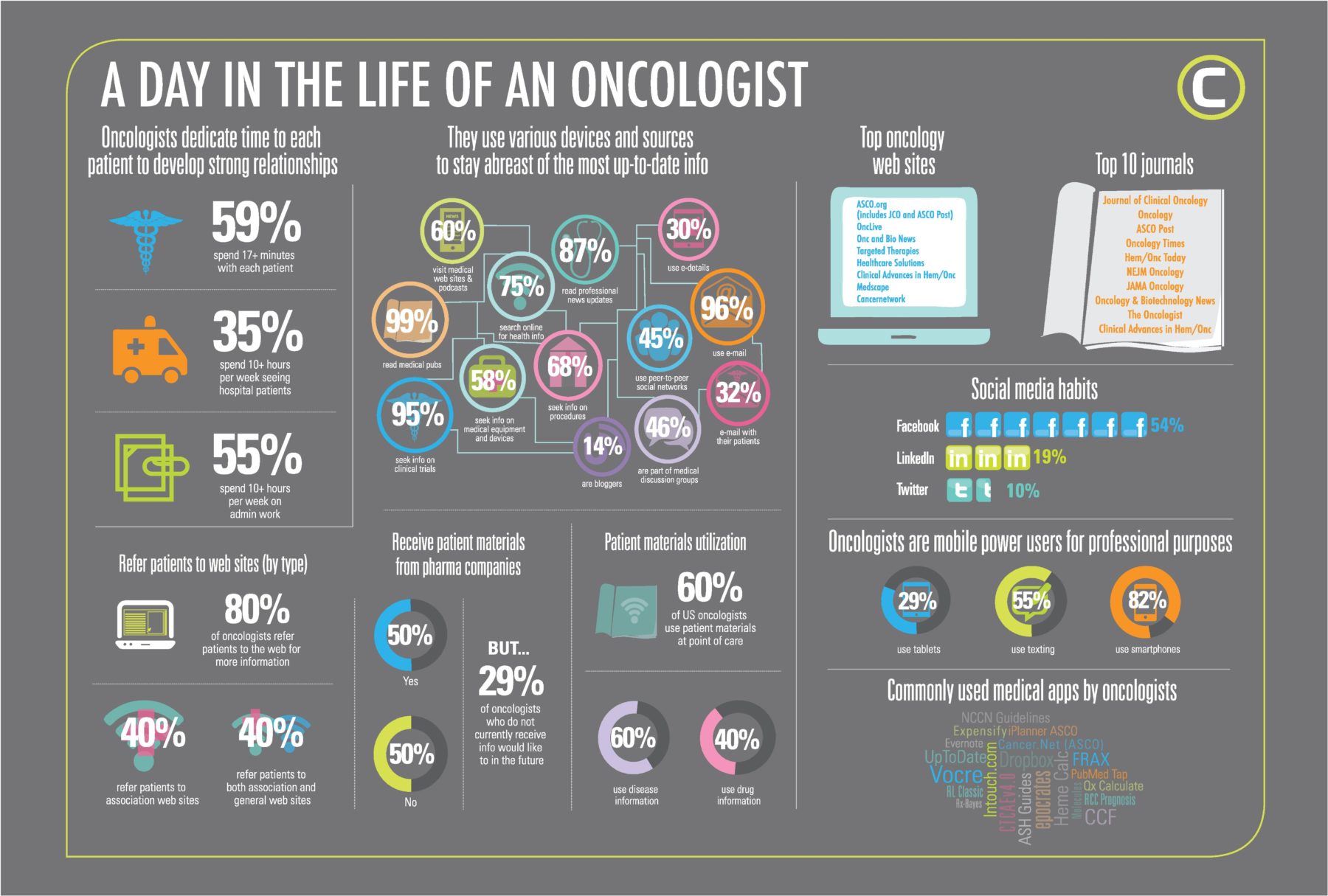
Web Exclusive: What You Need To Know About Oncologists
This infographic from Concentric Health Experience reveals the daily habits of oncologists and providers marketers with insights on where it is best to reach them.