There is no question that prior authorization (PA) requirements around prescribed branded and specialty medications are a major hurdle for patients and healthcare providers (HCPs). This fact has been a concern for years and is only continuing to grow: 51% of physicians report that PAs have significantly expanded in the last five years.
According to a survey by the American Medical Association (AMA), the increase in PAs alone has led 92% of clinicians to report patient care delays. Further, 75% of prescribers indicate that the PA process has led to their patients abandoning treatment altogether.
At best, a delay in approval of even a few hours postpones access to treatment and increases the risk of abandonment. While many, including the AMA, are actively calling for the reformation of the PA system to help reduce administrative burdens on healthcare providers and decrease waiting time for patients, it would be ill-advised to assume this is a change that will happen any time soon, if ever.1
In light of this, what can brands do to grow their profitability and market share, while supporting patient adherence and maintaining their first-choice status among HCPs? Is it wise to focus solely on the fight against expanding PA requirements, or can there be another way to combat the issue simultaneously?
Electronic Prior Authorization Helps Speed PA Responses
For many patients, providers, and brands, a significant improvement in adherence can be attributed to the increasingly widespread use of electronic Prior Authorization (ePA). Whereas it could take weeks to receive a response to traditional PAs, ePAs can be determined in just a few minutes when requirements are submitted quickly and the decision is straightforward.
The challenge, however, arises when approval is not a simple matter. The retrospective ePA process, which triggers the need for a PA upon denial at the pharmacy, all too often leaves patients, pharmacists, and prescribers stuck in a sort of limbo due to uncertainty a PA is even being requested. Any administrative delay in the process—prescriber being unaware that a PA is needed, the pharmacy not knowing a PA is being pursued, the patient seeing the full cost of the medication—may leave the patient with no choice but to walk away without their prescription. In other words, the increasing implementation of the ePA process certainly helps, but it is not enough to solve the PA dilemma.
So when brands are challenged with the task of supporting patient adherence while also maintaining profitability and market share, the answer may lie in finding innovative ways to use the ePA system to ensure patients leave the pharmacy with their medication in-hand. Recent case studies conducted by ConnectiveRx point to a comprehensive prescription first fill buy-down copay program built into the ePA process as a potential solution to this growing problem.
Prevent Patients from Leaving the Pharmacy Empty-Handed
It is widely understood that patient savings programs greatly increase access and adherence for branded and specialty medications. In fact, recent market research shows an 11.5% script lift when patient savings options are offered.2
However, most savings programs are generally available for approved medications, not pending ones. If brands offered to buy down the first fill of a medication before approval is received, the approval process could play out behind the scenes while patients are sent home with their medications. Such a program gives prescribers the time they need from an administrative perspective to provide adequate information to the payers to help secure approvals. If the process results in approval, the brand will be reimbursed for the first fill buy-down cost by the health insurer, and patients are more likely to refill and adhere to their medication, resulting in continued revenue for the brand.
Speaking of administrative burdens, this in itself is a major hurdle for HCPs and their staff. In fact, 64% of providers report altering their choice of brands due to access challenges, including PA.3 Additionally, if they believe brands are only offering copay cards after a medication is approved, there is little motivation for them to suggest their first choice when the risk of prescription abandonment is so high.
Mitigate Risks Around First Fill Buy-Down Programs
A program such as this is not without its risks. What happens, for instance, if a payer denies the PA regardless of everyone’s best efforts? A proper risk assessment analysis would help mitigate fears around buy-downs before approval and highlight success rates against inevitable denials. It is therefore imperative that brands acquire adequate data for each buy-down. Data transparency around approval and activity rates by prescription, pharmacy, geography, and payer are vital to ensuring a comprehensive understanding of the success of this program. Brands tracking activity and approval rates by prescriber can leverage this insight for more effective commercialization efforts.
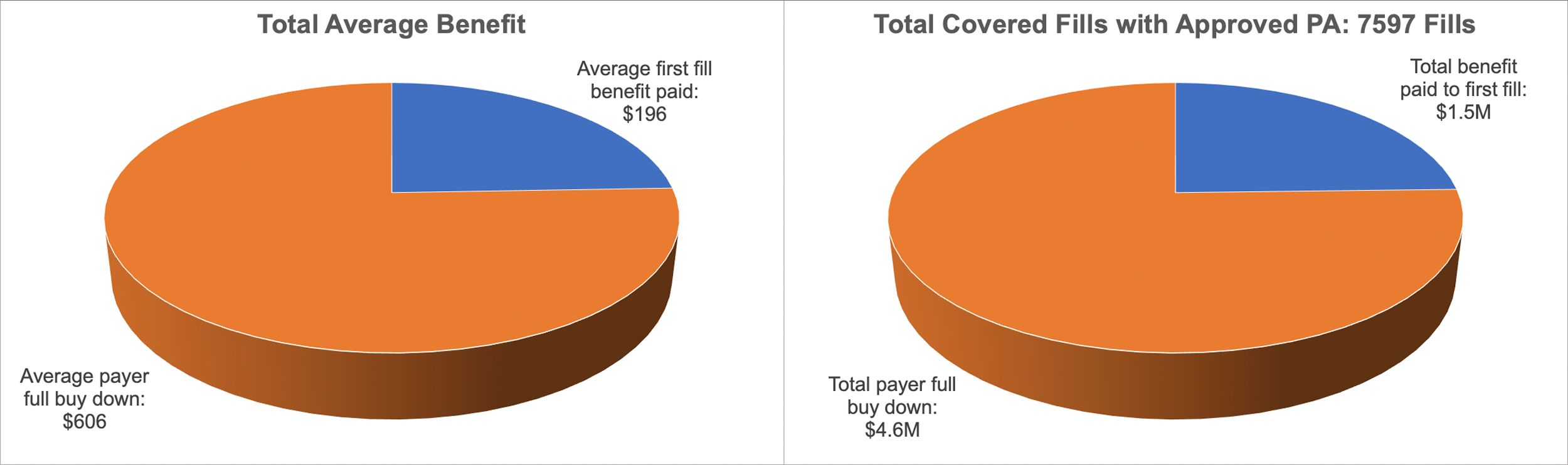
In the aforementioned case studies, ConnectiveRx did exactly this: Tracked ePA initiation status in order to determine cost vs. revenue. One brand (Brand X) offered a copay program which covered the cost of first fills while patients were awaiting PA decisions from their health insurers. Of those, 7,597 refills were subsequently covered as the PAs were approved. Brand X paid out an average of $606 for the buy down but just $196 for the refills. All told, the buy down program cost Brand X just short of $1.5M altogether; however, they earned more than $4.6M from payers, resulting in a revenue stream of more than $3M on refills alone (see Figure 1).4
 The Bottom Line
The Bottom Line
These trends in PA are not going away—almost all new brand launches are for specialty medications, after all—so brands should consider innovative, outside-of-the-box thinking that focuses on the big picture around revenue for the life of the script. Remember, the most important factors for overall patient health and brand revenue is to ensure patients are walking out of the pharmacy with their prescriptions in hand. Providing adequate time that allows the administrative hurdle to be overcome while the patient immediately initiates therapy is a reasonable path forward if you know your data and target opportunity well.
References:
1. https://www.ama-assn.org/practice-management/sustainability/prior-authorization-practice-resources.
2. Source: ConnectiveRx EHR analysis of 27 PhysicianCare with patient savings programs run between January 1, 2016 and December 31, 2018. Measurement is based on de-identified prescription data (written); range 0.09% to 29.05%. The results presented are representative of these programs only; each product situation is unique and future results may differ.
3. Source: ConnectiveRx, Patient price transparency research, Nov 2019. n = 70 prescribers exposed to standard insurance scenario ($80 patient out of pocket cost for [blinded medication]).
4. Source: ConnectiveRx, Case study for Brand X July 2019 – November 2019; January 2020 – April 2020.









