As is often the case, it’s not a single driver that shapes a trend but rather a confluence of many. Market Drill-Down takes current market data and slices it in new ways to reveal what’s really driving the market. It has been more than three months since Lipitor, the best-selling drug of all time, went off patent and began competing head-to-head with the first crop of generic atorvastatin. It’s no secret that Pfizer has been aggressively working to retain Lipitor’s brand share, through rebates, discounts and other strategies to improve reception by managed care and maintain loyalty with patients who now have lower-cost choices.
Pfizer has employed several strategies, including aggressive rebating with payers and co-pay cards to reduce patient out-of-pocket costs. With the co-pay card, patients are able to obtain their Lipitor prescription for as little as $4, which is the same co-pay as many generic products. While rebating and co-pay card strategies are pretty common today, Pfizer’s approach is more aggressive than most. Rather than targeting the discounts at certain audiences, Pfizer has made them available to virtually any eligible patient. As a result, in the month after generic atorvastatin became available, the average amount paid by patients for a Lipitor prescription dropped by 18 percent.
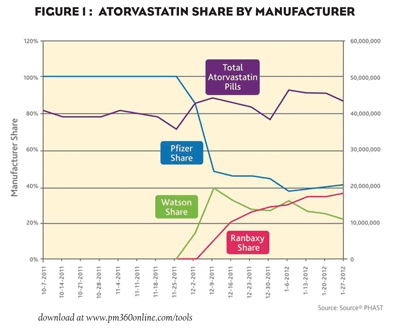
At face value, one might say Pfizer’s strategy is working. According to a recent analysis done by the inThought research team at Wolters Kluwer Pharma Solutions, 10 weeks after hitting the patent cliff, Lipitor holds an impressive 41 percent share of the atorvastatin market compared to 37 percent for Ranbaxy’s generic and 22 percent for Watson’s authorized generic (see Figure 1). As inThought points out, this is better than Aricept and Cozaar, two other blockbuster drugs that lost patent protection in the last couple of years. Most brands, in fact, capture less than 10 percent of the market after their patents expire. One might conclude that Pfizer is ahead of the game and well on its way to holding a substantial share of the atorvastatin market. But, if we look beyond what traditional prescription data tell us, we see several factors in the managed-care arena that weigh heavy on Pfizer’s effort. Using Source share. Dynamic Claims, which compiles data on the financial transactions that transpire between the pharmacies and the managed care plans each time a prescription is submitted, we see that, among the top commercial payers, only 35 percent of dispensed atorvastatin claims are for brand-name Lipitor. The rest are being captured by generics. The fact that the top nine payers— which control access to the vast majority of all patients in the U.S.—are working to switch patients to generic atorvastatin does not bode well for Pfizer’s ability to hold onto its current 41 percent share.
A SPIKE IN REJECTIONS AND REVERSALS
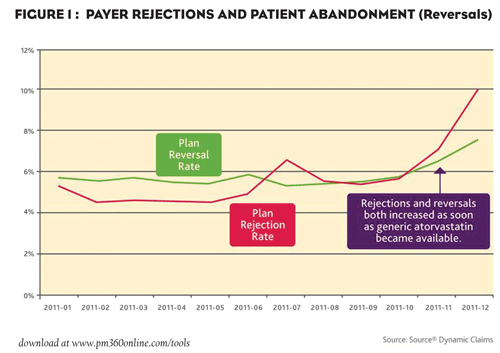
According to the claims data, payer rejections for Lipitor increased immediately after the availability of generic atorvastatin, running nearly 8 percent in December (see Figure 2). In addition, patient reversals also increased to nearly 10 percent. (Reversals happen when a patient abandons a prescription at the pharmacy, typically because the co-payment is too high.) As a result, “a sharp increase in both plan rejection rate and patient reversal rate for top Lipitor payers shows that plans are moving aggressively to migrate patients to generic atorvastatin,” according to inThought.
PFIZER NOW LARGEST U.S. PAYER FOR LIPITOR
When we dig deeper, we can see that among the non-government-dispensed claims, the Lipitor co-pay card paid for 24 percent of the payer-approved prescriptions in December. This little feat made Pfizer that month’s largest single “payer” for branded Lipitor. It is clear that holding onto market share is becoming a highly expensive undertaking.
IS ALL THIS HULLABALOO REALLY WORTH IT?
Pfizer is slowing the generic erosion of Lipitor, but will its effort help the bottom line? As inThought notes, with all of these costs adding up, “even if it sells more pills, its profit-per-pill is declining significantly.” We will need to watch the data carefully, to see if this strategy pays off. In the end, will Lipitor be any different from any of the other blockbusters that have hit their patent cliffs?
To download Market Drill-Down Tools for this issue’s topic, go to www.PM360online.com/tools
GET THE MARKET DRILL-DOWN EXPERTS WORKING FOR YOU! Submit your marketing question to Wolters Kluwer Pharma Solutions for deep analysis. All queries will be considered for a future column. Email Paula Fullman at Paula.Fullman@source.wolterskluwer.com