‘UID Plague’ and the ‘Hydra Effect’ weaken multi-channel marketing with errors that propagate through the entire data system. Avoid them with solid data design from the get-go.
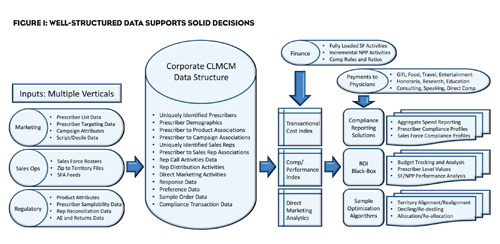
Multi-Channel Marketing (MCM) is a highly fashionable moniker often used too loosely to describe any initiative combining more than one avenue of promotion. In fact, though, best-of-breed MCM is a powerful and complex optimization of the available interactions between the pharmaceutical provider and its customers. MCM does not just facilitate messaging. It helps both manufacturer and consumer choose the best path through the shifting labyrinth of marketing options. MCM thrives on a level of complexity where consumers have multiple information options, and the manufacturer needs accurate information and insight on the consumer’s actions and interactions. Yes, MCM can be a data generating behemoth. Taming the monster by organizing the data structure is the real key to “demystifying” MCM.
Today’s pharma marketing budgets—best thought of as investments rather than expenses—often exceed 25% of the sales dollar. 1 The activity spans a vast geography of media: print, journals, websites, podcasts, email, social media, television, radio, face-to-face, and so much more. That’s the “multi” in multi-channel, and it’s on everybody’s mind, capturing attention not only in marketing, but in sales operations, regulatory compliance, IT, and finance. As it morphs from a marketing tactic to an integrated strategic initiative, the expectations and complexity expand.
THE NEW PARADIGM
Today, factors like downsizing and budget constraint foster an environment of increased reliance on non-personal promotion, and advances in technology broaden the opportunity to increase market share. At the same time, new regulatory requirements—like aggregate spend reporting—create additional costs by necessitating more granular visibility and tighter process control. Pharma is being squeezed right to left by regulation while being compressed top to bottom by economics. Though this “perfect storm” of pressure certainly drives demand for more flexible marketing solutions, it is not in and of itself responsible for MCM’s most recent buzz. Rather, the recognition of how all of these critical factors connect has catalyzed a re-imagining of MCM. We are beginning to understanding how each factor may depend on and complement the others. And how the data derived from a well-managed and integrated MCM framework can simultaneously serve a multitude of high level corporate goals.
If the enterprise can integrate the data-streams from all of its sales and marketing related channels within a skillfully crafted data structure, the resulting aggregate becomes a wellspring of strategic information, conjuring the shadow of an earlier practice, “Closed Loop Marketing” (CLM), which became popular in the 90’s as a method of keeping sales and marketing in sync through the sharing of centralized activity data. And it is CLM principles that may very well bridge today’s MCM with the broader multi-silo initiatives being conceived.
This hybrid, Closed Loop MCM (or CLMCM), creates the perfect environment for leveraging an extensive array of business activity information, supporting both ROI analysis and the prescriber-level reporting demanded by new regulation. Ideally, the system integrates inputs from “all” marketing and sales activity, allowing for assignment and tracking of related costs, and further incorporates results information such as marketing preference and script data to form a value matrix.
THE PERILS OF POOR DESIGN
That model is the shining prospect. However, CLMCM includes a little cloud with the silver lining. And generally, the root of the problem is a poorly designed data structure.
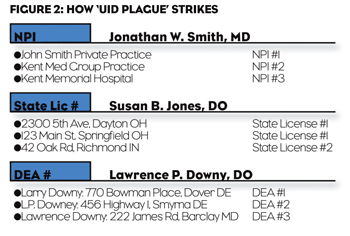
The UID Plague. Implementations often stumble at the fundamental first step: uniquely identifying prescribers. Doing this properly is surprisingly difficult, and without it, the system breaks down quickly. This is the “UID Plague.” The weaker the unique identification (UID) system, the faster and farther the system spirals into chaos.
For example, duplicate records generate duplicate, and sometimes conflicting, messaging. The system inundates both currently engaged prescribers and prospects with mixed messages from both personal and non-personal channels, leaving them confused and frustrated. Aggregate spend data becomes murky, inaccurate, and incomplete. Reports misrepresent the situation to marketing managers and don’t fully comply with regulatory requirements, despite the organization’s best intentions. Costs increase as marketing pays for redundant activities. ROI analysis becomes more convoluted, less efficient and less effective. The current field of prescriber data is littered with inconstant quasi-UIDs assigned by isolated systems and ad hoc suppliers. Stop-gap UIDs include the IMS number, State License Number, Drug Enforcement Agency (DEA) Number, and National Provider Identifier (NPI). Over-reliance on such UIDs, within the corporate master, can pollute the CRM and propagate inconsistency throughout the closed-loop process.
Examples: A physician can practice in more than one state, with two perfectly valid state licenses (and, likely, two different but perfectly valid addresses and, perhaps, two different versions of the physician’s name). If we simply look at the two state license records, we may not know it’s the same person. Proprietary solutions claim to identify the provider uniquely, but using them (with their contractual restrictions on use) limits the organization’s ability to deploy the identifier across the broad corporate master. NPI is problematic as it may be assigned to an “organization” as well as a prescriber. Resulting script data associated at the org level requires even more complex matching. In addition, NPI numbers can be assigned to non-prescribers, which can lead to unqualified records within the CRM.
The Hydra Effect. Named after the beast from Greek mythology, the Hydra Effect runs neck and neck (no pun intended) with the UID plague. It results when the Closed Loop isn’t really quite closed, and different organizations start grafting their own systems onto the whole. Pretty soon, you have a multi-headed Hydra preying on your MCM initiative, with each loosely coupled marketing effort working more or less independently. Acting unilaterally, with a narrower field of vision, they are unable to take advantage of the other groups’ insights, and can’t share their own. Like the UID Plague, the Hydra Effect causes disorganization, duplication of effort, and lost opportunities. It frustrates management, burns through budgets, and creates gaps in reporting data. One of the most common catalysts of the Hydra Effect is a poor, often incomplete, data structure. And the data structure is the core foundation of every CLMCM initiative. As you would expect, a weak foundation will ultimately cause catastrophic failure of whatever is built upon it.
Remember: Process relies on structure. You cannot maintain adherence or achieve true integration if the data structure does not facilitate and maintain complete and understandable information upon which to determine and coordinate your actions.
DEMYSTIFICATION—A NATURALLY OCCURRING BYPRODUCT
Data management and unique identification of prescribers are both art and science. Good CLMCM system development is founded on a rock-solid unique identification system, and incorporates state-of-the-art matching engines from its inception. It is not always a simple task to evaluate the ultimate integrity and completeness of a data structure. Expert advice (in or out of house) is a highly advisable first step in creating or improving a CLMCM model.
So, demystifying multi-channel marketing is more than possible. It is, in fact, a naturally occurring byproduct of a well-designed data structure and properly implemented closed-loop process. As we’ve seen, this sounds simple, but requires very thoughtful planning and meticulous attention to detail at the data level. If there is an ultimate key to successful MCM, that key is forged from data, and the door it opens leads to a carefully crafted and comprehensive closed-loop environment.
REFERENCE
1. Gagnon M-A, Lexchin J (2008) The Cost of Pushing Pills: A New Estimate of Pharmaceutical Promotion Expenditures in the United States. PLoS Med 5(1): e1. doi:10.1371/journal.pmed.0050001






