More than 20% of online conversations are health related. While the percentage of online conversations involving health may be high, this presents a unique challenge to health marketers in the social media space. Content in social media tends to be consumer driven and spontaneous in nature, a strong contrast to the control of the traditional Internet presentation of information through static web. The uncertainty lies in the ability, or rather the potential inability, to capture all adverse events on sponsored sites and the required information needed to create a complete report of an adverse experience.
Some of the difficulties in reporting are identifying patients, identifying reporters and pinpointing the suspect drug or biological product. Additionally, determining adverse experiences or fatal outcomes suspected to be due to the named drug or biological product can be challenging. Undeniably, the uncertainty related to the implication of the FDA regulations are limiting the social media potential within the industry and possibly creating a “tug of war” between legal and regulatory authorities and marketers.
Part of the challenge of monitoring social media for adverse events is turning large amounts of information into useful data. For instance, using pharma-specific social media analytics software to capture adverse events during a 12-month social and digital data analysis for a leading type 2 diabetes drug, 11,232 posts were found on the company’s brand-sponsored pages, excluding Wikipedia. Two hundred and eleven of the 11,232 posts reported as adverse events (1.8%). Such automated tools and processes allow an inside team to review about four pre-qualified posts a week, rather than manually reading through more than 30 posts a day for adverse events. Based on experience with many of the largest pharmaceutical companies, they have found it is important to go beyond the traditional definition of social media when ensuring that brands are in compliance with adverse event reporting.
Social Media and Web
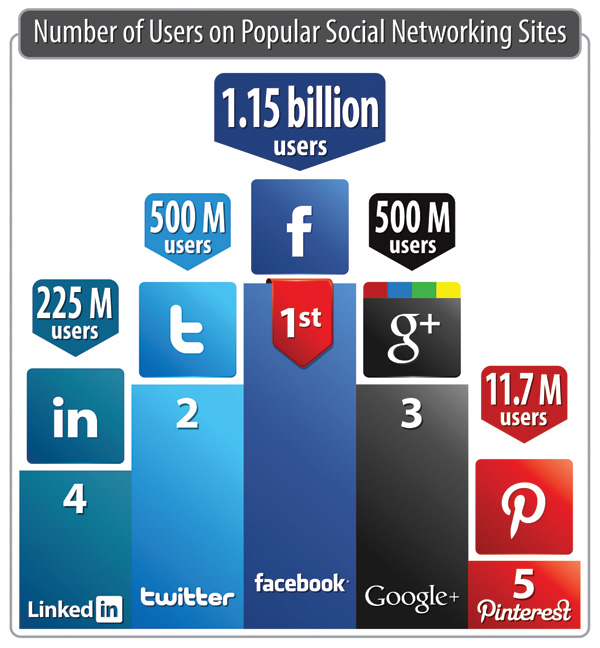
According to the digital analytics company comScore, there are only six countries where Facebook is not the dominant social networking tool (see Figure 1 below). The power with social media in healthcare is the ability to engage with your consumers in a meaningful way, while ensuring that the proper adverse event reporting processes are in place, as manual monitoring sometimes is not sufficient to support the volume of comments received. In addition to the more obvious channels online, it is important to review comments in YouTube, branded product review functionality, etc.
Mobile
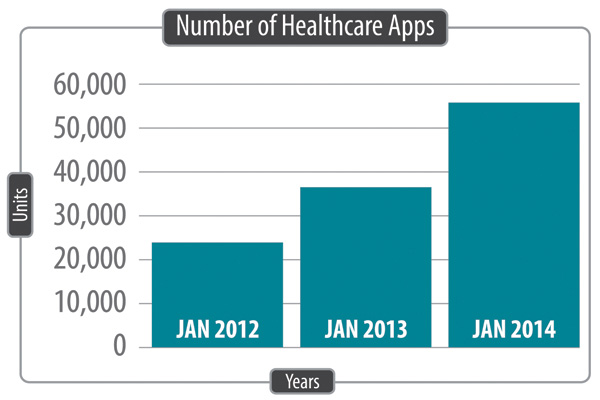
The forecast for healthcare-related mobile apps is anticipating strong growth in the number of applications to be launched over the next two years (see Figure 2 below). And recently companies have been focusing their efforts to ensure that consumer comments in the online mobile application store are monitored for adverse events.
Any Electronic Record
With ongoing enhancements in business productivity tools, it is important to evaluate current internal adverse events monitoring and tracking processes to include larger initiatives such as CRM, EMR and market research. Any report, comment or note with a mention of an adverse event needs to be evaluated and reported to the FDA according to regulatory guidelines.
Social media is enabling connections and engagement, while success is tied to the perceived value among key audiences. With the spontaneous nature of social media content, automated adverse event processes with an appropriate layer of human validation are increasingly important to support a compliant ready environment optimized for real-time engagement and growth.