FROM THE COCHRANE DATABASE OF SYSTEMATIC REVIEWS
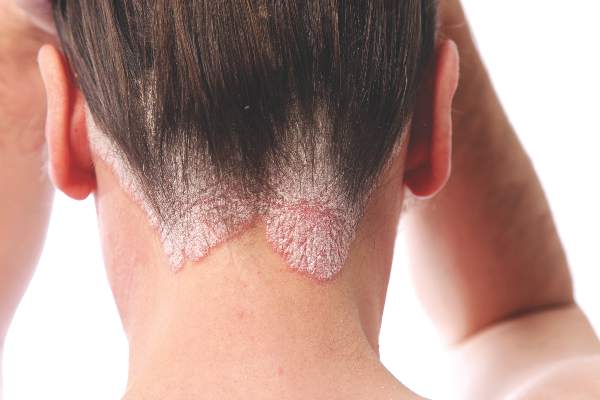
As a treatment for scalp psoriasis, the combination of a topical steroid and topical vitamin D was marginally better than topical steroids alone, but both approaches have a similar safety profile, according to a Cochrane review.
The systematic review included 59 randomized controlled trials of topical treatments for scalp psoriasis, representing a total of 11,561 participants of all ages, most of whom were followed for less than 6 months, according to Justin Schlager, MD, of the Charité – Universitätsmedizin Berlin, and coauthors (Cochrane Database of Systematic Reviews 2016. Issue 2. doi: 10.1002/14651858.CD009687.pub2 ).
In the analysis, topical steroid monotherapy was significantly better than topical vitamin D for clearance, as assessed by the Investigator’s Global Assessment of Disease Severity (risk ratio, 1.82; 95% confidence interval, 1.52-2.18). But the combination of a topical steroid and vitamin D showed a small but statistically significant advantage over steroids alone (RR, 1.22; 95% CI, 1.08-1.36) and an even greater advantage over vitamin D alone (RR, 2.28; 95% CI, 1.87-2.78).
Similarly, for treatment response, the combination of a topical steroid and vitamin D showed the greatest benefit in terms of treatment response when compared with steroid monotherapy (RR, 1.15; 95% CI, 1.06-1.25) – an additional benefit the authors observed was small – and when compared with vitamin D alone (RR, 2.31; 95% CI, 1.75-3.04).
But for monotherapy, corticosteroids were more than twice as effective as vitamin D alone for treatment response (RR, 2.09; 95% CI, 1.80-2.41). The analysis also showed that corticosteroids of moderate, high, and very high potency were similarly effective.
Steroids were associated with a significantly lower risk of withdrawal because of adverse events than vitamin D (RR, 0.22; 95% CI, 0.11-0.42). Patients using topical steroids reported adverse events such as a burning sensation or irritation at the site of application, while patients taking vitamin D reported side effects such as pruritus, candidiasis, dermatitis, and erythema, although in both cases these were mostly limited to the application site.
The combination of vitamin D and topical steroid had a lower risk of withdrawals because of adverse events than vitamin D alone, but showed a similar rate to topical steroids alone.
“Given the similar safety profile and only slim benefit of the two-compound combination over the steroid alone, monotherapy with generic topical corticosteroids may be fully acceptable for short-term therapy,” the authors wrote. The authors noted that data on patients’ quality of life was poor across all the studies included in the analysis and called for future trials to address this gap, and more long-term assessments.
“Regardless of the type of psoriasis, up to 79% of people with the condition present with scalp involvement, which has frequently been the first site to show symptoms of the disease,” they pointed out.
Thirty of the 59 studies were either conducted or sponsored by the manufacturer of the study medication, and the authors described the overall quality of the studies as “moderate.” Thirty-three studies were double blind, 14 were single blind, two had “third-party” blinding, six were open-label, and four did not report blinding information.
The study was supported by the Universidade Federal de São Paulo, Brazil; the Universidade Federal do Rio Grande do Norte, Brazil; and the National Institute for Health Research, United Kingdom. Six authors and one clinical referee declared speakers’ fees, research grants, and funding from the pharmaceutical industry. One author had no conflicts of interest to disclose.